Dche.coe.upd.edu.ph
Department of Chemical EngineeringCollege of Engineering
University of the Philippines
Diliman, Quezon City
Laboratory Safety Manual 2015
TABLE OF CONTENTS
Emergency Information:
Emergency Telephone Numbers
Basic First Aid
First Aid Responder
First Aid Boxes
Emergency (Fire) Alarms
Coping with an Emergency
Health and Safety Procedures in the ChE Department
Risk Assessments
Local Rules and Advice:
Preventing Fires and Floods
Personal Safety
Safety Checklist for Laboratory Users Personal Protective Equipment Laboratory Chemical Fume Hood Chemical Storage Cabinets Safety Showers Eyewash Stations Safety with Chemicals Safety with Electricity Safety with Machines and Transport Safety with Glassware Safety with Refrigerators, Freezers & Cryogenics Safety when Heating and Cooling Down Noise and Safety Outside Regular Working Hours Unattended Experiments Pregnancy Tidiness Reporting Accidents and Incidents Chemical Waste Disposal Security in the Department Emergency Telephone Numbers
UP DILIMAN POLICE /FIRE UP Health Service/Ambulance 928 3608 (Direct) 981 8500 local 2701, 2702, 2704 Nearest Hospital (East Avenue Medical Center) Bureau of Fire Protection Quezon City Hall 928 8363 / 924 1922 Philippine National Red Cross Quezon City Chapter 433 2151 to 52 / 434 3751 436 0948 / 924 3101 MMDA (Office of Public Safety) 882 0851 / 882 4151 to 70 local 374 Campus Maintenance Office (Quick Response Team) ChE Department Office 981-8500 local 3113-3114 Basic First Aid
If anyone falls unconscious following an injury, get the attention of the security guard on-
duty. If after office hours, notify the UP Diliman Police at 928 3615.
Cuts
Control the bleeding by applying pressure over the wound with a pad of paper tissue or towel
roll.
Do not attempt to remove any broken glass, splinters, etc.
Seek medical attention. Avoid contact with blood.
Burns
Douse the burn with copious amounts of cold water (e.g., under a running tap).
Do not attempt to remove anything sticking to the burn.
Seek medical attention.
Chemicals
On Skin: Wash off with copious amounts of water. Seek medical attention as needed.
In Eyes: If available, use an eye-wash spray with the eyelids held open. Otherwise use a cold
running tap. Seek medical attention as needed.
Inhaled: If possible, move to fresh air. Seek medical attention.
Swallowed: Seek medical attention. Identify the chemical.
Electricity
Switch off power. Get the attention of the security guard on-duty for help.
Other injuries - falls, blows
Call for medical attention.
In the event of a serious injury, use the emergency number 928 3608 which accesses the UP
Health Service, or 928 3615 which accesses the UPD Police both on a 24-hour line.
For minor injuries, First Aid Responders may be called.
Name of First Aid Responder
Local No.
Julie Anne del Rosario
Diwata Ronda
Karl Ezra Pilario
Myron Alcanzare
First Aid Boxes
First Aid Boxes should be kept stocked according to the list posted on the front or within the box. All First Aid material should be available from the Department of Chemical
Engineering Administrative Office. Each Research Laboratory Head is to ensure that the
stock of the First Aid box is regularly maintained.
Emergency (Fire) Alarms
The buildings in the Department of Chemical Engineering are equipped with a fire detection
system that will sound the alarms if a sensor detects flame, heat or smoke, or if the pull lever
alarm is activated.
Note that false alarms may sound due to ageing or defective fire detectors. In the event of an
alarm, spend a few seconds only in turning off heating equipment or making your
experiment safe before leaving the building by the route that has been described to you.
Assemble at the designated muster area for a roll-call.
Do not attempt to enter the building until you have been told by any member of the Disaster Risk
Reduction and Management (DRRM) Committee that it is safe to do so. The members of the
DRRM are as follows:
Asst. Prof. Julie Anne del Rosario Engr. Karl Ezra Pilario Engr. Myron Alcanzare Engr. Charlimagne Montealegre Mr. Mark Batadlan Coping with an Emergency
Regular Working Hours: These are from 8:00 AM to 5:00 PM Monday to Friday. Know at least two routes from your workplace to an Exit.
Know the locations of Telephones, Fire Extinguishers and
Blankets, Fire Alarm Points, Safety Showers, Eye Wash Stations
and First Aid Boxes.
Know how to contact Campus Security.
Know how to switch off electricity, gas, water, etc. in and around
your workplace.
.
Evacuation Procedure All evacuation drills will be notified in advance (except for the exact time). In the event of an evacuation, follow the route given in the
Department of Chemical Engineering Emergency Evacuation
Route map. Assemble away from danger and clear of the building.
Do not re-enter the building until you are told that it is safe to do so.
If the fire is clearly minor, deal with it using an extinguisher but do
not endanger yourself. Normally the sensors will detect any fire and
the Alarm will operate automatically. Otherwise, operate the nearest
Alarm Point and call 928 3615 for UPD Police.
Leave the area immediately. Close all doors on exit. Operate the
nearest Fire Alarm Point. Warn people to avoid the affected area
and call 928 3615 for UPD Police. Aiding an injured Person Either phone for a First Aid Responder or if more serious, call 928 3615 for UPD Police, then a First Aid Responder. Even if the
Alarms sound, do not attempt to move the injured person out of
the building unless there is imminent danger. Stay with the
injured person until help arrives. If the injury is slight they should be
taken by taxi with a First Aid Responder in attendance, or if the
injury is more serious by Ambulance.
If the leak is obvious, stop it; call 928 3615 for UPD Police. Warn
people in labs below to safeguard papers and equipment. Do not
attempt to move wet electrical equipment until it is disconnected
from the mains.
Failure of Mains Service Failure of fume-hood, gas, water, electricity during working hours, look for the Building Administrator (local 3113-3114). Outside the regular working hours, call Campus Security 928 3615. Health and Safety Procedures in the Department of Chemical
Engineering
The online version of the DChE Health, Safety and Environment Management System may be
found a
People who work only in an Office or who do only Computing need not study every aspect
discussed in this handbook. But those working in Laboratories, Boiler Rooms, and/or
Machine shops must take the time to read the Safety Manual and take to heart the
information therein on Health and Safety procedures in consideration of the greater potential
hazards in said areas.
Risk Assessments
If you are a faculty member, research staff, or laboratory staff, you should read the information
that follows and that applies to different classifications of personnel.
Risk assessments are procedures aimed at estimating risks to health and safety associated with
any activities one performs. Having estimated the risks, workers are expected to devise rules and
protocols to minimize identified risks. Safety-compliant chemical engineering plant practice
requires that risk assessments be carried out for all work that will be done, and that workers are
informed of the outcome of the assessment, and trained to carry out their duties safely. The Risk
Assessment procedure applies to all kinds of work activities.
If you are working only in a regular office
If you are part of the clerical staff or a researcher doing a project which is purely computational, then the only risk assessments you need to read are the following: General Office Work Visual Display Equipment Standard Electrical Equipment If you are a Researcher, you will need to put in writing that you have read these assessments by filling up the appropriate form and submitting to the Office of the HSE Officer. If you are working in a Laboratory
If you are working on a research project, your supervisor or project leader should supply you with a copy of your Project Risk Assessment. This will give you guidance regarding the risks you must be aware of and be trained to handle. A good reference for prepared risk assessments on the use of typical laboratory equipment, for chemicals and materials may be found in while the ChE Department is preparing its own. After going through the prepared risk assessments, workers are to complete a comprehensive activity-based risk assessment (LAB FORM 3) for their particular work, considering every step in their activity. Preventing Fires and Floods
Each laboratory must have a laboratory door sign posted outside each lab, containing the names and contact information of persons to contact in case of an emergency. The sign must also identify the type(s) of hazards to be expected in the lab. Fire: Fires can result in disfiguring and disabling injury or death. A fire may consume or
destroy all your work, leaving you with nothing to continue on.
The Fire Detection System: The Department of Chemical Engineering Building is equipped
with a fire detection system. A red light appears/blinks when a detector is activated. When the
alarm sounds, you must leave the building before the alarm is over.
Take care NOT to cause false alarms. However, if you happen to cause a false alarm, inform the
Department Office at 981-8500 loc 3113-3114 immediately.
Solvents in the Laboratories. Only a working minimal amount of flammable solvents may be
stored in any laboratory. Excess amounts of solvents in a room could endanger life and property
in case of fire. Typical regulations recommend a maximum quantity in liters of flammable
liquids stored in any one laboratory to be 1.6 x floor area of the lab in square meters. As much
as possible, especially overnight, solvents should be stored in the ventilated safety cabinet
provided for the purpose.
Leaving a Laboratory or Machine Shop: When leaving the workplace for the night, or when
going away for a long time even during the day, workers have the responsibility to ensure that:
there are no obvious problems with reactions or equipment left running (these should be properly labeled) unnecessary electrical equipment, e.g., ovens, is turned off and no naked flame or flammable gas is left on. flammable solvents are properly stored in closed storage cupboards. fume-hoods are closed lights are turned off fire doors and other doors are closed.
Floods: Floods damage equipment, paperwork and fixtures. Floods can also be dangerous by
causing ceiling boards to loosen up and fall or causing electrocution by wetting live electrical
equipment. Floods should be avoided.
Water Cooling Connections: Plastic/rubber tubings that carry cooling water to any set-ups
must be fastened securely onto the apparatus and the water taps using suitable clamps, wires,
tags or clips. The exit tube must deliver the water down a drain that is large enough to
accommodate the flow without overflowing. The exit tube must likewise be securely anchored
to prevent splashing or ejection in case water pressure rises.
PERSONAL SAFETY
Safety Checklist for Laboratory Users1
1. Do you wash your hands before leaving the laboratory? 2. Do you wear appropriate attire in the laboratory (lab coat, safety googles, 3. Are the appropriate hazard signs and emergency numbers posted on the outside of the laboratory door? 4. Are all containers in your lab properly labeled? Do you know how to interpret 5. Do you know where to find Material Safety Data Sheets (MSDS's) for all chemical used in your laboratory? 6. Are the chemicals in your lab properly stored? 7. Have you been instructed in the proper use and handling of the chemicals in your laboratory? 8. Have you been instructed in the location and use of safety devices (safety showers, eye washes, laboratory hoods, etc.) in your laboratory? 9. Are you aware of emergency procedures in the event of a chemical exposure, spill, fire, or explosion? 10. Do you know how to properly segregate and dispose of chemicals that you 11. Are all mechanical engineering controls (e.g., fume hoods) operating 12. Do you know whom to notify if working in a laboratory alone after normal 13. Have you attended the Laboratory Use Orientation and Safety Seminar conducted by the Department? 14. Have you attended laboratory specific training provided by your laboratory 15. Do you know where your laboratory Standard Operating Procedures (SOP's) is stored and how to use them? 16. Are you familiar with the protocol that you are following? Do you feel comfortable performing the procedure without additional information or demonstration? If you answered NO to any of these questions, read this laboratory safety manual. If you still have questions, see your laboratory supervisor or contact DChE at 981 8500 loc 3113/3114. PERSONAL BEHAVIOR
Do not prepare, store (even temporarily), or consume food or beverages in any chemical Avoid wearing or bringing lab coats outside the laboratory. Do not smoke in any chemical laboratory. Additionally, be aware that tobacco products in opened packages can absorb chemical vapors. Do not allow visitors, including children and pets, in laboratories where hazardous chemicals are stored or are in use, or hazardous activities are in progress. Confine long hair and loose clothing while performing experiments. Do not apply cosmetics when in the laboratory. Do not use mouth suction to pipette chemicals or to start a siphon. Use a pipette bulb or a mechanical pipetting device to provide a vacuum. Do not distract or startle other workers. Wash well before leaving the laboratory. Keep work areas clean and free from obstruction. Do not block access to exits, emergency equipment, controls, electric panels, etc. Do not working alone. Avoid loitering, especially when conducting experiments.
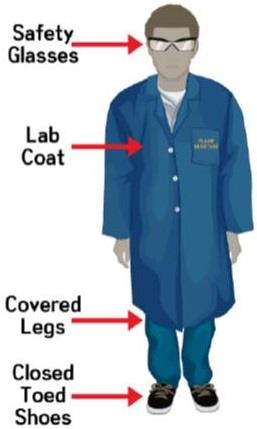
PERSONAL PROTECTIVE EQUIPMENT2
Personal protective equipment (PPE) is a special gear used to protect the wearer from specific hazards of a hazardous substance. It is a last resort protection system, to be used when substitution or engineering controls are not feasible. PPE does not reduce or eliminate the hazard, protects only the wearer, and does not protect anyone else.
Figure 1. Personal protective equipment3
PPE includes protective clothing, footwear, eye protection, gloves, and respiratory protection. The need for PPE is dependent upon the type of operations and the nature and quantity of the materials in use, and must be assessed on a case by case basis. Workers who rely on PPE must understand the functioning, proper use, and limitations of the PPE used.
Table 1: Personal protective equipment
Protective Clothing
When the possibility of chemical contamination exists, wear
protective clothing that resists physical and chemical hazards over your casual clothes.
Lab coats are appropriate for minor chemical splashes and solids
contamination, while plastic or rubber aprons are best for
protection from corrosive or irritating liquids.
Wear closed-toes shoes at all times in the laboratories where
chemicals are stored or used or equipment are being operated.
Use chemical-resistance overshoes or boots when handling
corrosive chemicals or large quantities of solvents or water that might penetrate normal footwear.
The following are strictly not allowed:
× open-toed shoes
× high heeled shoes
× stiletto-heeled shoes
× backless shoes
Wear safety glasses whenever you are performing experiments,
especially those involving chemicals, pressure apparatus, or mechanical equipment.
Do not wear contact lenses.
Wear protective gloves when handling hazardous materials,
chemicals of unknown toxicity, corrosive materials, rough or sharp-edge objects, or very hot or very cold materials.
Make sure to use the gloves appropriate for the type of work that
you will be doing. You may refer tofor a few glove materials and their general uses; the list is not exhaustive.
While wearing gloves, be careful not to handle anything aside from
the materials in your procedure. Touching equipment, phones, wastebaskets, or other surfaces may cause contamination.
Do not wear rubber or plastic gloves when working with a naked
Before removing them, wash the outside of the glove. To avoid
accidental skin exposure, remove the first glove by grasping the cuff and peeling the glove off the hand so that the glove is inside out. Repeat this process with the second hand, touching the inside of the glove cuff, rather than the outside. Wash hands immediately with soap and water.
Respiratory protection Use a respirator when engineering controls (e.g., general ventilation,
fume hood) are not feasible or do not reduce the exposure of a chemical to acceptable levels.
Table 2: Glove materials and their general uses
Glove Material
General Uses
Offers the highest resistance to permeation by most gases and water vapor. Especially suitable for use with esters and ketones.
Provides moderate abrasion resistance but good tensile strength and heat resistance. Compatible with many acids, caustics and oils.
Excellent general duty glove. Provides protection from a wide variety of solvents, oils, petroleum products and some corrosives. Excellent resistance to cuts, snags, punctures and abrasions.
Provides excellent abrasion resistance and protection from most fats, acids, and petroleum hydrocarbons.
Highly impermeable to gases. Excellent protection from aromatic and chlorinated solvents. Cannot be used in water or water-based solutions.
Exceptional resistance to chlorinated and aromatic solvents. Good resistance to cuts and abrasions.
Resists a wide variety of toxic and hazardous chemicals. Provides the highest level of overall chemical resistance.
Provides flexibility and resistance to a wide variety of acids, caustics, salts, detergents and alcohols.
LABORATORY CHEMICAL FUME HOOD
Chemical fume hoods capture, contain, and expel emissions generated by hazardous chemicals. Some fume hoods channel captured air from the room to the outside, while other fume hoods capture only the pollutants and return the air to the room. In general, it is advisable to conduct all experiments involving chemicals in a fume hood. Fume hoods offer an extra measure of protection, especially for experiments that involve the release of undesirable or hazardous effluents.
Table 3: Laboratory chemical fume hood
Use the fume hood only when you are familiar with its
Maintain the sash at or below the optimum operating height
as designated by the label with an arrow.
Raise large objects that must be in the hood (e.g., water bath)
to allow airflow beneath and on all sides of the object.
Always work back into the hood, six inches beyond the sash
line, keeping the sash line between your body and your work.
Keep the inside of the hood clean and uncluttered.
Make sure that the hood is always in good condition and
capable of routine use.
If you detect strong odors released from materials in the
hood, check to make sure that the ventilation fan is on.
If the fume hood is malfunctioning, discontinue work and
inform lab head, UR, or supervisor.
Aside from the standard PPE, wear a face shield or explosion
shield when handling reactive substances.
Prevent any solid objects or materials from entering the
exhaust ducts to avoid clogging.
Do not use the fume hood for long-term chemical storage.
CHEMICAL STORAGE CABINETS
Storage of flammable and corrosives in the lab should be limited to the least possible quantity. Flammable materials should be stored in properly constructed wooden or metal storage cabinets. Storage cabinets for flammable materials shall be conspicuously labeled, "Flammable - Keep Fire Away."
Table 4: Chemical Storage Cabinets
Use and Maintenance
Do not arrange chemicals in alphabetical order without
consideration of their chemical compatibilities.
Limit storage outside of the cabinet to materials that are in
Store glass containers in the bottom shelf of storage cabinets.
SAFETY SHOWERS
Safety showers provide an effective means of treatment in the event that chemicals are spilled or splashed onto the skin or clothing.
Safety showers should be in a clearly marked location. The facility should be no more
than 10 seconds away from every laboratory workbench.
Laboratory workers should be able to locate the nearest shower with their eyes closed. Safety showers should be able to supply a continuous stream of water to cover the entire
Safety showers should be located away from electrical panels or outlets. If at all possible, safety showers should be installed near appropriate drainage system.
Table 5: Safety Showers
Stand below the showerhead and pull down the ring chain or
triangular rod to activate the shower.
Remove contaminated clothing, including shoes and jewelry,
while under an operating shower.
For the laboratory personnel: Drain and test eye wash
stations weekly, and inspect every six months.
EYEWASH STATIONS
Eyewash stations provide an effective means of treatment when chemicals come in contact with eyes.
The eyewash facility should be clearly marked and no more than 10 seconds away from
every lab work station.
Laboratory workers should be able to locate the nearest eye wash facility with their eyes
An eye injury is usually accompanied by a skin injury. For this reason, eye wash stations
should be located near safety shower and/or drench hose so that the eyes and body can be washed.
Table 6: Eyewash stations
Use and Maintenance
Aim water or eye solutions at the base of the nose between
the eyes, not directly into the eyeball. This increases the chance of effectively rinsing the eyes free of chemicals. Harsh streams of water may drive the particles further into the eyes.
You may need to forcibly open the eyelids to attempt eye
Flood your eyes and eyelids with water or eye solution for a
minimum of 10 minutes.
For the laboratory personnel: Drain and test eye wash
stations weekly, and inspect every six months.
SAFETY WITH CHEMICALS
It is mandatory for safety to do a health risk assessment prior to handling chemicals. Follow MSDS recommendations and heed warnings regarding the use and storage of chemicals. As a matter of safety, MINIMIZE the amount of reactants used in an experiment. SUBSTITUTE hazardous chemicals as much as possible. The following are emphasized: Common Solvents
Are they toxic? Are there flammability or explosion danger limits on vapor concentration in air?
Use an effective fume-hood particularly in handling volatile solvents. Fine powders or substances that release dust during handling can be toxic and explosive!
Use absorbent granules, sawdust or sand to mop up solvent spills. When using large amounts of acid or base or any amount of strongly smelling material, keep a neutralizing agent within reach.
Category 1 – known carcinogens to humans. Warning Label: Toxic Category 2 – strongly presumed carcinogen, based on long-term animal studies and other information. Risk Phrase: R45 (May cause cancer) or R49 (May cause cancer by inhalation) Category 3 – possibly carcinogenic. Warning Label: Harmful. Risk Phrase: R40 (Limited evidence of a carcinogenic effect). When working with Category 1 or 2 carcinogens, worker must be fully informed of the grave cancer risk and the procedures for proper handling and disposal. Project Leader or Laboratory Head must keep a record of all user exposure to carcinogens. Disposal: DO NOT dispose along with the waste solvents. SPECIAL ARRANGEMENTS must be made by the project leader/lab head with a chemical waste disposal company.
Because mercury is VERY TOXIC, all glass apparatus containing mercury
should be provided with a secondary containment to catch mercury in the event of a breakage. If there are mercury-containing items in the lab, have a MERCURY SPILL KIT (sulfur powder and suction devices) on hand. Other things that may be used to clean up mercury include: latex or vinyl gloves, zip-lock bags or plastic trash bags, wide tape (masking, duct, or clear), paper towels, eyedropper, two index cards or pieces of stiff cardboard, water to moisten the paper towels. Mercury spills should be collected up immediately before they roll into cracks, crevices, or get into clothing, shoes and other porous materials from which it would be very difficult to remove. Avoid spreading spilled mercury! Do not use brooms, mops or vacuum cleaners to clean up a mercury spill; cordon off the spill area so nobody walks through it, and take off any contaminated clothing, shoes, and other items and leave it in the spill area (do not bring them to other areas). Contaminated clothes, rags, carpets, shoes, and other porous items will likely need to be disposed as hazardous waste – wrap these in double plastic trash bags, carefully seal with tape, and label. Small spills (e.g., from a thermometer or thermostat) –
For liquid mercury spills, refer to
For shattered fluorescent bulbs and compact fluorescent bulbs, see
Large spills – EVACUATE the spill area; SEEK professional help (fire department or a contractor). TEST clothing and personal belongings that may have been contaminated. Refer to the following webpage:
Cyanides MAY NOT BE USED outside regular working hours.
(highly toxic) Hydrofluoric acid HF MAY NOT BE USED outside regular working hours. (highly corrosive)
HF can cause very painful burns at concentrations above 1M or 2% in water without immediately observable effects. When working with HF, have a tube of HF Antidote Gel/Calcium Gluconate at hand, apply if concentrated acid contacts the skin. Inhalation of anhydrous HF can be fatal.
Liquid Nitrogen can cause severe burns upon eye or skin contact. Use safety goggles and gloves when handling cryogenic liquids. Be wary of explosions due to sudden expansion of condensed gases or vapors. Highly reactive substances (e.g., sodium metal, metal hydrides) – quench in a fume hood in the absence of water or solvents.
(e.g. Acetone, ethyl acetate, ethanol, hexane, etc…)
Store in Safe containers inside cabinets designed for flammables (fire resistant). NEVER heat flammables using an OPEN FLAME. When using flammables, KEEP AWAY from IGNITION SOURCES. ALWAYS use a FUME HOOD while working with flammable liquids.
(e.g., Peroxides, nitrates, nitrites, perchlorates, chlorates, chlorites,
hypochlorites, dichlorates.) STORE AWAY from flammables, organic compounds and combustible materials STORE AWAY from processing and handling areas. STORE AWAY from other materials especially organic or reducing agents. STORE in glass or other inert containers, and use compatible secondary trays. For larger containers, provide dikes around storage areas and ramps at door openings. STORE below eye level to reduce risk of accidental dropping of containers. Some oxidizing agents (e.g. 8% or higher H2O2 in water) generate oxygen gas and water during storage need to store in vented containers. DO NOT STACK vented containers on top of one another. STORE away from exits. Perchloric acid may only be used in a fume hood designed for perchloric
acid. Purchase or use of anhydrous perchloric acid in the ChE department is forbidden.
(e.g., Sulfuric acid, chromic acid, stannic acid, hydrofluoric acid, NH4OH) STORE separate from bases and flammables. Many acids are also strong oxidizers Add acid to water, never the other way around. Store corrosives BELOW or AT EYE LEVEL. USE SECONDARY containers for storage to contain leaks. STORE in a WOODEN CABINET or one with corrosion-resistant coating. Store NITRIC ACID in a separate cabinet of compartment.
Water-reactives – store and isolate away from any water source
Phosphorics – store in a clearly marked, isolated part of the lab
Peroxide-forming (e.g. ethers, acetals, ketals, cyclic ethers, aldehydes,
benzylic hydrogens, allylic hydrogens, vivinyl, vinylidene compounds, dienes) – should be labelled for date received and expected shelf life. NEVER OPEN any chemical containers if peroxide formation is suspected, or if cover is stuck up. Visually inspect for crystals and unusual viscosity before opening.
Shock-sensitive - (e.g., trinitro glycrine, …. see Appendix D for a more
complete list of shock-sensitive materials). Store in explosion-proof
containers where there is minimal human activity and little risk of getting
unnecessarily disturbed. Be wary of bottles or containers with signs of
crystal formation which may indicate peroxide formation. Do not force-
open these, DO NOT shake the bottle.
SAFETY WITH ELECTRICITY
Electrical hazards can cause burns, shock and electrocution. Never assume that a wire is safe to touch! Never operate electrical equipment while you are standing in water, or when your feet are soaking wet. If you are not an electrician, do not attempt to repair electrical cords or equipment. Always use caution when working near electricity. Notice the danger When using any electrical equipment, WATCH for worn out cables and signs
insulations, loose connections, loose plugs, etc. RECTIFY or REPLACE immediately when necessary.
and Wet electrical equipment is VERY DANGEROUS. DISCONNECT from
the mains before touching it.
Have portable electrical equipment tested regularly by an authorized electrician. New, second-hand, and old equipment must be tested before being used. Clearly mark failed equipment as "FAILED" and do not attempt to use.
SAFETY WITH MACHINES AND TRANSPORT
Carrying solvents
Observe safety when transporting Winchester or reagent bottles of solvents by using appropriate carriers or trolleys.
Rotary equipment Be wary of getting one's hair, clothing, wires or tubing caught in rotating
Use appropriate trolleys in transporting large cylinders of compressed gas. Gas cylinders should be securely strapped at all times (during transport, storage and use). Keep only the minimum number of gas cylinders in a room or station. On the gas regulators, mark the maximum safe pressure allowed for regular use in the laboratory.
SAFETY WITH GLASSWARE
Glassware under vacuum poses implosion risks. In the event of an
Pressurized glass
implosion, glass pieces may be sprayed around COVER any evacuated glass with strong sticky tape, plastic net, or a metal shroud. Glassware under pressure is MORE DANGEROUS. Use only with TOTAL CONTAINMENT.
Broken glass and Put broken glassware ONLY in dedicated waste bins labelled "SHARPS", empty bottles
which should be taped up at all sides and bottom before use. All bottles for disposal should be inspected and treated prior to disposal:
Remove and destroy any sodium residues found in bottles, then
wash the bottles with water.
Completely empty all bottles of solvents and chemicals, wash and
dry them until there is no residual odor in them.
Put cleaned bottles without lids or stoppers into the waste bins
All glassware for repair should be thoroughly cleaned first. Use appropriate
degreasers as needed.
SAFETY WITH REFRIGERATORS, FREEZERS & CRYOGENICS
Refrigerators and
Check contents frequently and regularly purge of unwanted samples.
Use only an explosion-proof refrigerator for storing chemicals. Never store food in a refrigerator used for chemicals. Tightly seal all containers that are put in the refrigerators and freezers.
Nitrogen Be aware of the danger of asphyxiation in closed environments (elevators,
air-conditioned laboratories, storage rooms, etc.) when boil off occurs. A block of dry ice can completely sublime and reduce oxygen levels to
suffocation level in an elevator. Use stairs where possible, or transport without humans in the elevator. Beware of sudden boil-off of O2 that condenses from air due to low temperatures.
SAFETY WHEN HEATING AND COOLING DOWN
Use Heating jackets or oil baths. Due to the danger of fire and explosion, DO NOT use ovens or furnaces for heating autoclaves that contain organic solvents. Where such reactions are needed, perform in a fume hood labelled adequately with the contents of the reaction vessel and the explosion risk.
Heated sealed tubes or bottles should be allowed to cool completely to room temperature before opening.
NOISE AND SAFETY
The sense of hearing will be able to detect a change in operation of an equipment or machinery earlier that the sense of vision. Try to keep background noise (from pumps, shakers, compressed air jets, etc.) at lowest level possible so as not to impair hearing. The use of personal audio equipment (e.g., radios and earphones) is NOT allowed.
OUTSIDE REGULAR WORKING HOURS
When working outside of the regular working hours, there are additional risks involved since help may not be available in the event of an accident. All research advisers should be aware of the work being undertaken by their students. They should also ensure that work outside the regular hours is properly regulated. For work outside of regular lab hours:
Undergraduates are forbidden to be in the laboratories unless a supervisor is present with
Working alone is NOT allowed. Make sure there is always someone within earshot.
Experiments that have a risk level beyond 3 must be performed only during the regular
working hours. No work involving cyanides or HF may be done outside normal work hours.
UNATTENDED EXPERIMENTS
Experiments left running unattended pose special risks in terms of fires and floods and must be carefully controlled. The following must be observed:
Unattended running of experiments may be carried out only when absolutely necessary.
Experiments involving overnight refluxing of solvents must be within a ducted fume
hood. All water lines must be securely fastened.
All experiments left running must have a notice on it
o stating EXPERIMENT IN PROGRESS, PLEASE LEAVE RUNNING
o and indicating POTENTIAL HAZARDS in understandable language (e.g.,
Flammable Solvent, Contains Toxic Material). Give Filipino translation as well.
o the name and telephone number of the person responsible for the experiment.
This telephone number must be accessible and one which is with the concerned person at all times.
o If at all possible, the above information must be posted in a prominent position
outside the laboratory.
o Keep the light of the fume hood ON if the set-up is in a fume hood. o Electrical equipment left on should be labeled "DO NOT SWITCH OFF" o For large, permanent equipment (e.g., SEM), post contact numbers outside the
laboratory and clearly identify EMERGENCY ELECTRICAL OFF switches.
PREGNANT AND BREASTFEEDING WOMEN
Pregnant and breastfeeding women must inform their adviser in writing in order to avoid exposure to agents or tasks that may endanger the unborn child or infant.
TIDINESS
Keep your working area reasonably tidy and free of obstacles to minimize risks of accidents, fires, and floods.
Reporting Accidents and Incidents
Any dangerous event in the Department ChE Laboratories must be reported immediately to the HSE Officer or the Assistant Chair, to avoid recurrence of said event. Dangerous incidents are unplanned events in which no one was hurt, but which had the potential to cause injury or damage to property.
Accidents are defined as events in which someone gets hurt. All incidents, accidents and even near-misses must be reported to the HSE Officer or Assistant Chair. Please note that the aim is NOT to apportion blame for an accident or incident, but rather to put up measure to prevent recurrence. No one should be afraid to report such events. It is mandatory in industry practice.
Chemical Waste Disposal
Environmental regulations require proper disposal of chemical waste. Such entails
additional cost and inconvenience, hence as a matter of practicality and economics, it is
good practice to design experiments such that waste generation is minimized. As much
as possible and practicable, substances or materials should be recovered and recycled.
At the conclusion of research projects, workers must dispose of all unwanted products or
other chemicals, including leftover unused reagents. Proper chemical waste disposal is a
requirement for laboratory clearance.
Waste Chemicals
As much as possible, the lead researcher of individual or group research project is to
make arrangements with a chemical waste disposal company for disposal of unwanted
chemicals. A list of such companies may be obtained from the Environmental
Management Bureau of the DENR.
All substances to be disposed of should be identified by chemical name and molecular
formula. For mixed waste, the character of the mixture must be accurately defined (e.g.
organic amines and their salts, no compound boiling below 100 C, some suspected
carcinogens). Descriptions that are not acceptable are "mixture of organic liquids",
"smells of nitrobenzene". IMPORTANT: known hazards associated with the water
should be clearly stated on the label, including hazardous drying agents.
Container must be leak-proof, clearly labeled with identity or category of the contents,
any known hazards, some indication of the boiling point range. Trade names alone are
not acceptable. The chemical nature must be indicated.
If packed into used boxes, ENSURE OLD LABELS ARE ERASED OR
OBLITERATED. Also, mark the container as to which SIDE UP. Total weight should
NOT exceed 5 kg and the dimensions should be about one foot cube. Organic or aqueous
liquids should be in glass or plastic containers, solids in metal drums or plastic tubs.
Waste Solvents
Prior to disposal, submit a form listing the type and quantity of waste to be disposed.
There are 2 categories of waste solvents¨ Chlorinated and non-Chlorinated. Put only
approved waste solvents (with limited amounts of solute at worst) in the containers. DO NOT put reaction mixtures containing drying agents, oxidants or solutions of oxidants must never be put into the waste solvent containers. Acids and bases must be placed in separate containers, never into containers for organic solvents. No substance that is category 1 or 2 carcinogens may be put in the waste solvents in any form. For details on waste disposal and classification of chemical waste, please see the Waste Management System of the ChE Department.
Security in the Department
All students, faculty and staff should wear their ID's at all times when within the premises of the building. All visitors and guests should log in at the security guard's logbook at the lobby, and must wear their visitor's pass at all times. All doors and windows should be locked when no one is in the lab or office. Be alert for suspicious persons (e.g. non-students without visitor's passes) in the premises, especially outside regular working hours. Ask what he/she wants. Contact security personnel or the UP Police at 928-3615 if unsure about the person's intention. Observe greater caution when working outside regular working hours. Report to the security personnel any person within the premises who does not bear an authorization pass. Suspicious movements, especially in areas immediately surrounding the building, should also be alerted to security personnel. For added security, CCTV cameras are installed in various locations. For research laboratory heads, regularly update the roll call or list of persons authorized to work in your lab, and post the list on a prominent location outside your lab (e.g. door).
First version authors:
Dr. Rizalinda de Leon
Asst. Prof. Patrick Ramoso
Asst. Prof. Marjorie Baynosa
Asst. Prof. Julie Anne del Rosario
Engr. Jonas Karl Christopher Agutaya
Engr. Joseph Jimievard Abuan
Engr. Myron Alcanzare
Acetylene -- flammable Acetyl peroxide -- flammable Ammonium picrate -- requires >10% water to reduce shock hazard Benzoyl peroxide -- requires 25%-35% water to reduce shock hazard Bouin's solution -- yellow solution containing picric acid Chlorotrifluoroethylene -- flammable Dinitrophenylhydrazine -- requires >10% water to reduce shock hazard Dipicrylamine (syn: Hexanitrodiphenylamine) -- explosive and poisonous Dipicryl sulphide (syn: Hexanitrodiphenyl sulfide) -- requires >10% water to reduce shock hazard Nitrofluoride -- strong oxidizer Nitrogen trifluoride (syn: Methyl ethyl ketone peroxide) -- poisonous gas Nitroglycerin -- skin irritant Nitromethane (syn: Nitrocarbol) Picramide (syn: Trinitroaniline) Picric acid (syn: Melinite, Trinitrophenol) -- requires >10% water to reduce shock hazard Picryl chloride Picryl sulphonic acid (syn: trinitrobenzenesulfonic acid) -- flammable solid, corrosive Trinitroanisole (syn: trinitrophenylmethyl ether) -- explodes by heat or shock Trinitrobenzene -- requires >10% water to reduce shock hazard Trinitrobenzoic acid -- an explosive Trinitrotoluene (syn: Picryl sulphonic acid, TNT) -- moderate explosion hazard; requires strong
Urea nitrate -- requires >10% water to reduce shock hazard
Source: http://dche.coe.upd.edu.ph/wp-content/uploads/2015/05/04-UPD-ChE-Laboratory-Safety-Manual.pdf
Progestin-only pills for contraception
Progestin-only pills for contraception (Review) Grimes DA, Lopez LM, O'Brien PA, Raymond EG This is a reprint of a Cochrane review, prepared and maintained by The Cochrane Collaboration and published in The Cochrane Library2013, Issue 11 Progestin-only pills for contraception (Review)Copyright © 2013 The Cochrane Collaboration. Published by John Wiley & Sons, Ltd.
Microsoft word - lecture_no._15_gerd_tw_25052008.doc
Lecture no. 15 Esophageal Motility Disorders - 3 Gastroesophageal Reflux Disease This chapter could begin like this: the esophageal mucosa is unable to endure contents coming from the stomach. These contents may be fluids secreted by the stomach itself (the most oft-occurring possibility), or even duodenal-biliary in origin. These events lead to a anatomical-clinical condition that generally goes by the name of esophagitis.