Ri.conicet.gov.ar
5834 • The Journal of Neuroscience, March 27, 2013 • 33(13):5834 –5842Central Dopamine D2 Receptors Regulate Growth-Hormone-
Dependent Body Growth and Pheromone Signaling to
Conspecific Males
Daniela Noaín,1* M. Ine´s Pe´rez-Milla´n,2* Estefanía P. Bello,1 Guillermina M. Luque,2 Rodrigo Casas Cordero,1
Diego M. Gelman,1 Marcela Peper,1 Isabel García Tornadu,2 Malcolm J. Low,3 Damasia Becu´-Villalobos,2
and Marcelo Rubinstein1,4
1Instituto de Investigaciones en Ingeniería Gene´tica y Biología Molecular, and 2Instituto de Biología y Medicina Experimental, Consejo Nacional de
Investigaciones Científicas y Te´cnicas, C1428ADN Buenos Aires, Argentina, 3Department of Molecular and Integrative Physiology, University of Michigan
Medical School, Ann Arbor, Michigan 48105, and 4Facultad de Ciencias Exactas y Naturales, Universidad de Buenos Aires, C1428EGA Buenos Aires,
Argentina
Competition between adult males for limited resources such as food and receptive females is shaped by the male pattern of pituitary
growth hormone (GH) secretion that determines body size and the production of urinary pheromones involved in male-to-male aggres-
sion. In the brain, dopamine (DA) provides incentive salience to stimuli that predict the availability of food and sexual partners. Although
the importance of the GH axis and central DA neurotransmission in social dominance and fitness is clearly appreciated, the two systems
have always been studied unconnectedly. Here we conducted a cell-specific genetic dissection study in conditional mutant mice that
selectively lack DA D2 receptors (D2R) from pituitary lactotropes (lacDrd2KO) or neurons (neuroDrd2KO). Whereas lacDrd2KO mice
developed a normal GH axis, neuroDrd2KO mice displayed fewer somatotropes; reduced hypothalamic Ghrh expression, pituitary GH
content, and serum IGF-I levels; and exhibited reduced body size and weight. As a consequence of a GH axis deficit, neuroDrd2KO adult
males excreted low levels of major urinary proteins and their urine failed to promote aggression and territorial behavior in control male
challengers, in contrast to the urine taken from control adult males. These findings reveal that central D2Rs mediate a neuroendocrine-
exocrine cascade that controls the maturation of the GH axis and downstream signals that are critical for fitness, social dominance, and
competition between adult males.
appreciated in mice lacking D2Rs (Drd2 ⫺/⫺) that display hyper- Central D2 receptors (D2Rs) participate in important brain func- prolactinemia and develop pituitary hyperplasia tions, including the control of locomotor activity and reward- Analysis of Drd2 ⫺/⫺ mice also revealed the seeking behavior. D2Rs are also found in pituitary lactotropes, unexpected importance of D2Rs in the regulation of the growth where they mediate the tonic inhibitory control that dopamine hormone (GH) axis and control of body size, a differential phe- (DA) exerts on prolactin (PRL) synthesis and release. The notype that is considerable in males physiological significance of this inhibitory control has been Drd2 ⫺/⫺ mice display a shortfall of pituitary somatotropes, re-duced GH and IGF-I serum levels, and are dwarfs The abnormally large lactotrope/somatotrope ratio Received Dec. 12, 2012; revised Jan. 24, 2013; accepted Feb. 22, 2013.
observed in Drd2 ⫺/⫺ mice is highly reminiscent of a similar phe- Author contributions: D.N., M.I.P.-M., E.P.B., G.M.L., R.C.C., D.M.G., M.P., I.G.T., D.B.-V., and M.R. designed nomenon observed in lactating females, in which suckling- research; D.N., M.I.P.-M., E.P.B., G.M.L., R.C.C., D.M.G., M.P., I.G.T., and M.R. performed research; M.J.L. and M.R.
contributed unpublished reagents/analytic tools; D.N., M.I.P.-M., E.P.B., G.M.L., R.C.C., D.M.G., M.P., I.G.T., D.B.-V., induced inhibition of tuberoinfundibular DA neurons reduces and M.R. analyzed data; D.N., M.I.P.-M., M.J.L., and M.R. wrote the paper.
lactotrope D2R stimulation and promotes differentiation of so- This work was supported in part by the Howard Hughes Medical Institute (International Research Scholar Grant matolactotrope precursors into functional lactotropes to M.R.), Tourette Syndrome Association (to M.R.), Universidad de Buenos Aires (to M.R.), Consejo Nacional deInvestigaciones Científicas y Te´cnicas (CONICET, Argentina, to D.B.-V.), and the Agencia Nacional de Promocio´n Therefore, it is conceivable that Científica y Tecnolo´gica (to M.R. and D.B.V.). D.N., M.I.P.-M., E.P.B., G.M.L., D.M.G., M.P., and I.G.T., were recipients pituitary D2Rs play a critical role in establishing the terminal of doctoral fellowships from CONICET. We thank the National Hormone and Pituitary Program (National Institute of differentiation ratio of these two cell types from their common Diabetes and Digestive and Kidney Diseases) and Dr. A.F. Parlow for mouse RIA kits and antibodies, Guillermo Lanuza somatolactotrope precursor and, for providing Ai14 mice, and Vanina Rodriguez and Irina García Sua´rez for excellent technical assistance.
*D.N. and M.I.P.-M. contributed equally to this work.
therefore, in shaping the GH axis.
The authors declare no competing financial interests.
An alternative hypothesis that may account for the somato- Correspondence should be addressed to Dr. Marcelo Rubinstein, Vuelta de Obligado 2490, Buenos Aires 1428, trope shortfall and dwarfism of Drd2 ⫺/⫺ mice is that lack of Argentina. E-mail: [email protected].
central D2Rs impairs growth hormone-releasing hormone Copyright 2013 the authors 0270-6474/13/335834-09$15.00/0 (GHRH) or somatostatin (SST) function. In fact, the phenotype Noaín, Pe´rez-Milla´n et al. • Central D2Rs in Body Growth and Social Dominance J. Neurosci., March 27, 2013 • 33(13):5834 –5842 • 5835
observed in Drd2 ⫺/⫺ mice is similar to that present in lit/lit mice taining 2% normal goat serum (NGS); rabbit anti-rat adrenocortico- that lack functional GHRH receptors tropic hormone (rACTH; NHPP, NIH) diluted 1:1000 in KPBS/0.3% and in mutant mice lacking Ghrh Triton X-100 containing 2% NGS; and rabbit anti-LH (NHPP, NIH) because these two mouse models have fewer somatotropes diluted 1:12,000 in KPBS/0.3% Triton X-100 containing 2% NGS. Sec- and display dwarfism.
tions were then washed twice in PBS for 20 min at room temperature andincubated for 2 h with a secondary antibody at room temperature. To In the present study, we sought to determine whether GH detect GH, ACTH, and LH, a goat anti-rabbit Alexa Fluor 488 (Invitro- insufficiency in Drd2 ⫺/⫺ mice was due to the lack of D2Rs in gen) secondary antibody diluted 1:1000 in KPBS/0.3% Triton X-100 was pituitary lactotropes or in the brain by conducting a functional used; to detect prolactin, a donkey anti-goat Alexa Fluor 488 secondary dissection strategy based on cell-specific Drd2 inactivation in antibody diluted 1:500 in KPBS/0.3% Triton X-100 was used. Sections conditional Drd2 mutant mice We developed were washed twice in KPBS for 20 min and mounted in Vectashield a strain of transgenic mice expressing cre from a mouse prolactin (Vector Laboratories). Pituitary sections of neuroDrd2KO mice and gene promoter, Tg(Prl-cre) 1Mrub, that in parallel with transgenic control littermates were incubated with rabbit anti-GH antibody mice expressing cre from a rat nestin promoter, Tg(Nes-cre) 1Kln/J (NHPP, NIH) diluted 1:500 in KPBS/0.3% Triton X-100 containing 2% NGS. After washing twice for 20 min with KPBS, sections were incubated eliminate D2Rs selectively from pituitary lactotropes or from with donkey anti-rabbit FITC-coupled IgG (Santa Cruz Biotechnology)diluted 1:500 in KPBS/0.3% Triton X-100 for 2 h at room temperature.
cells of neural origin, respectively. Our results showed that al- After washing twice in KPBS, sections were incubated with propidium though male mice lacking D2Rs in pituitary lactotropes iodide to stain cell nuclei. Finally, sections were rinsed in KPBS twice for (lacDrd2KO) developed a normal GH axis and body size, those 10 min, mounted in water, and coverslipped using Vectashield (Vector lacking D2Rs in the brain (neuroDrd2KO) showed an impaired Laboratories). Confocal microscopy was performed to assess the number GH axis and reduced body growth. Deficits in the male pattern of of cells expressing GH per pituitary.
GH release are associated with feminization of sexually dimor- In situ hybridization. Brains and pituitaries were freshly removed and phic liver proteins, including major urinary proteins (MUP; frozen at ⫺40°C in isopentane, equilibrated at ⫺20°C in Tissue-Tek, a group of pheromones excreted at high levels in the cryosectioned at 16 m, and mounted on SuperFrost slides (Fisher Sci- male urine and used in male-to-male social interactions entific), and stored at ⫺70°C until use. Sections were thawed and main- Therefore, in this study, we also sought to tained at room temperature for 30 min and then fixed in 4% investigate whether neuroDrd2KO male mice exhibited deficits paraformaldehyde in PBS for 30 min. After two washes in PBS, slideswere incubated in 0.1 M triethanolamine, pH 8, for 3 min, followed by in MUP levels and if their urine failed to induce territorial and incubation with triethanolamine-acetic anhydride 0.0025% for 10 min.
aggressive behaviors in conspecific males.
Slides were then rinsed twice in 2 ⫻ SSC (0.3 M NaCl, 0.03 M sodiumcitrate, pH 7.2), and, finally, dehydrated quickly in ascending ethanol Materials and Methods
concentrations. Sections were incubated with hybridization buffer (66% Generation of mice lacking D2Rs in pituitary lactotropes. A transgene construct formamide, 260 mM NaCl, 1.3 ⫻ Denhardt's) for 1 h at 57°C and hybrid- carrying 2.9 kb of 5⬘ mouse Prl promoter sequence driving cre recombinase ization started after adding 5 ⫻ 10 6 to 10 7 cpm/ml of an antisense mouse expression with a coding sequence for a nuclear localization was used to Drd2 exon 2 [ 35S]-riboprobe and proceeded overnight at 57°C in a wet generate Prl-cre transgenic mice. The 4.2 kb Prl-cre transgene was microin- chamber. The next day, slides were washed 4 times with 4 ⫻ SSC and then jected into the pronuclei of fertilized B6D2F2 eggs. The selected line incubated with RNase A (20 g/ml in 0.5 M NaCl; 10 mM Tris-HCl pH 8; Tg(Prl-cre) 1Mrub was backcrossed for five generations to C57BL/6J.
1 mM EDTA pH 8) at 37°C for 30 min. Slides underwent consecutive Tg(Prl-cre) 1Mrub (n ⫽ 5) mice were consecutively crossed to Drd2loxP/loxP washes with 2⫻, 1⫻, and 0.5 ⫻ SSC/1 mM DTT and a final wash with mice to generate Drd2 loxP/loxP.Tg(Prl-cre) mice, named 0.1 ⫻ SSC/1 mM DTT at 65°C for 30 min. Finally, sections were dehy- lacDrd2KO. Transgenic mice were produced at the Instituto de Investigacio- drated in ascending ethanol concentrations, covered in Kodak N-TB2 nes en Ingeniería Gene´tica y Biología Molecular (CONICET, Argentina) and emulsion, and exposed for 15–20 d in a dark chamber until development animal procedures were conducted in accordance with local regulations and for posterior analysis. For the digoxigenin-labeled probe, prehybridiza- the Guide for the Care and Use of Laboratory Animals (National Institutes of tion was performed in a solution containing formamide 66%, NaCl 260 Health [NIH]).
mM, 1.3 ⫻ Denhardt's, EDTA 1.3 mM, Tris-HCl 13 mM, pH 8.0, and Generation of mice lacking D2Rs in neurons. To ablate D2Rs from dextran sulfate 13% at 57°C for 1 h. The probe was incubated at 65°C for cells of neural origin, Drd2 loxP/loxP mice were crossed to B6.Cg- 2 min in 0.5 mg/ml tRNA and 10 mM dithiothreitol and added to the Tg(Nes-cre) 1Kln/J (The Jackson Laboratory) to obtain cohorts of prehybridization buffer at a final concentration of 85 ng/ml. Hybridiza- Drd2 loxP/loxP (control) and Drd2 loxP/loxP.B6.Cg-Tg(Nes-cre) 1Kln/J tion was performed at 70°C overnight. After hybridization, the sections were washed with 0.2% SSC at 72°C for 1 h and then twice with a solution Immunofluorescence. Double transgenic mice, obtained by crossing containing 100 mM NaCl, 0.1% Triton X-100, and 100 mM Tris-HCl, pH Tg(Prl-cre) 1Mrub mice with the cre reporter mouse line Ai14 7.5. Sections were then incubated with the same solution plus 10% NGS received cabergoline (Laboratorios Beta) 0.5 mg/kg intraperi- for 4 h and then incubated with alkaline phosphatase-conjugated anti- toneally to increase prolactin signal and 2 h later were perfused with digoxigenin Fab fragments antibody (1:3500; Roche) at 4°C overnight.
paraformaldehyde 4% in phosphate buffered saline (PBS). Pituitaries The following day, samples were washed 5 times for 15 min each in PBS were incubated in perfusion solution at 4°C overnight, then in PBS for and then washed 4 times in a solution containing 100 mM NaCl, 50 mM 24 h, and finally in sucrose 10% in PBS for additional 24 h. Fixed pitu- MgCl , 0.1% Tween 20, and 100 m M Tris-HCl, pH 9.5. Then samples itaries were mounted in gelatin cubes (gelatin 10%, sucrose 10% in PBS) were incubated with NBT/BCIP (Roche) in the above-mentioned buffer and stored at ⫺80°C until use. Coronal 12 m sections were cut using a overnight at room temperature. Signal development was stopped with Leica CM1850 cryostat and mounted on Superfrost slides (Fisher Scien- PBS, coverslipped with Mowiol (polyvinyl alcohol, glycerol in Tris 0.2 M, tific). For prolactin and luteinizing hormone (LH) detection, an antigen pH 8.5) and photographed.
retrieval procedure was performed incubating the sections in sodium Binding autoradiography. Brain and pituitary sections were obtained as citrate buffer 10 mM, pH 8.5, at 80°C for 10 min. Incubation with primary described for in situ hybridization, except they were stored at ⫺20°C antibodies were performed overnight at 4°C. The primary antibodies until use. Slides were thawed and maintained at room temperature for 30 used were as follows: goat anti-prolactin (sc-7805; Santa Cruz Biotech- min, and then preincubated in binding buffer (50 mM Tris-HCl, 120 mM nology) diluted 1:200 in KPBS/0.3% Triton X-100 containing 2% normal NaCl, 5 mM KCl, 2 mM CaCl , 1 m , pH 7.4) for 1 h. Next, slides donkey antiserum; rabbit anti-hGH (National Hormone and Pituitary were incubated for 1 h in binding buffer containing 1 nM [ 3H]- Program [NHPP], NIH) diluted 1:1000 in KPBS/0.3% Triton X-100 con- nemonapride. Nonspecific binding was determined in the presence of 10 5836 • J. Neurosci., March 27, 2013 • 33(13):5834 –5842
Noaín, Pe´rez-Milla´n et al. • Central D2Rs in Body Growth and Social Dominance M S-(-)-sulpiride, and then sections were rinsed twice for 5 min in cold immobility for each mouse over the five trials. The tube test, performed water, completely dried with cold air, and exposed for 15–30 d to [ 3H]- to assess social dominance, consisted of introducing two adult mice in sensitive film (BIOMAX MR Scientific Imaging Film; Kodak) at room opposite entrances of a cylindrical acrylic tube of 3.4 cm diameter and 30 temperature in a dark cassette.
cm length and allowing them to interact in the center of the tube by Radioimmunoassays. Anterior pituitaries were dissected and homoge- removing a gate placed at 15 cm from each entrance. It is expected that nized in 0.2 ml of PBS. Protein contents were measured with the QUBIT one of the animals will force the other to retreat from the tube by pushing Fluorometer and the QUANT-IT protein Assay Kit (Invitrogen). Ali- it out. Successful trials were considered those in which a mouse retreated quots of equal quantities of protein were used to measure GH content by from the tube being pushed by the victor. We also observed atypical trials RIA. Serum PRL and GH pituitary content were measured by RIA using in which a mouse walked backwards by itself, not being forced by its kits provided by the National Institute of Diabetes and Digestive and opponent. Trials in which neither of the two mice pushed the other out Kidney Diseases (NIDDK; Dr. A.F. Parlow, NHPP, Torrance, CA). Re- during the 10 min test were considered null and discarded from the sults are expressed in terms of mouse PRL standard RP3 or mouse GH analysis. We assayed give pairs per genotype: C57BL/6J versus C57BL/6J standard AFP-10783B, respectively. Intra-assay and interassay coeffi- females, Drd2 loxP/loxP males, or neuroDrd2KO males. First, we trained all cients of variation were 7.2% and 12.8%, and 8.4% and 13.2%, for pro- mice to enter the tube by both entrances. After training, each pair of lactin and GH, respectively. For IGF-1 RIA, serum samples (15 l) and confronting mice was assayed three times in the tube and the 15 trials IGF-1 standards were subjected to the acid-ethanol cryoprecipitation considered in the analysis. A variant of the intruder-resident aggression method. IGF-1 was determined using antibody (UB2– 495) provided by test, using castrated wild-type (WT) mice as intruders, was used to deter- Drs. L. Underwood and J.J. Van Wyk, and distributed by the Hormone mine the aggressive display promoted by the fresh urine of Drd2 loxP/loxP or Distribution Program of the NIDDK. Recombinant human IGF-1 (rh neuroDrd2KO male mice on isolated sex- and age-matched C57BL/6J IGF-1; Chiron) was used for radioligand and unlabeled standards. The mice C57BL/6J adult male mice were isolated for assay sensitivity was 6 pg per tube. Intra-assay and interassay coefficients at least 1 month and then wild-type castrated adult male mice were of variation were 8.2% and 14.1%, respectively. For testosterone RIA, an swabbed on their genitals and back with 150 l of saline or fresh active testosterone RIA kit (DSL-4000; Diagnostic System Laboratories) collected urine from Drd2 loxP/loxP or neuroDrd2KOmice. After 1 min of drying, intruders were introduced to the home cage of an isolated Urine dosage of major urinary proteins. Urine was collected from male for 3 min. All tests were videotaped and analyzed for determi- 6-month-old mice between 1500 and 1700 h and centrifuged briefly for 3 nation of latency to first aggressive contact, consisting of kicking, min at 8800 ⫻ g. Two microliters of the supernatant was boiled in SDS biting, wrestling, or tumbling and total time spent attacking. The buffer. Samples were fractionated in 12% SDS-PAGE and subsequently odor-stimulated scent-marking test was performed using as experi- stained with Coomassie blue. The 20 KDa MUP represents the major mental subjects C57BL/6J adult male mice and, as stimulating odor protein component of mouse urine.
sources, the cages in which Drd2 loxP/loxP or neuroDrd2KO mice had Semiquantitative real-time PCR. Hypothalami from Drd2 loxP/loxP and been housed for the previous 48 h (test cages). The bedding of the test neuroDrd2KO mice were excised to determine Ghrh and Sst mRNA cages was removed but the cage itself was not cleaned. A rectangle (13 levels. Tissue samples were immediately homogenized in TRIzol reagent cm ⫻ 22 cm) of absorbent paper (Whatman, 3 mm Chr) was placed (Invitrogen), RNA extracted, and first strand cDNA synthesized using over the floor of each cage. Immediately, an adult male C57BL/6J and MMLV reverse transcriptase (Epicenter Biotechnolo- previously isolated for 1 month was placed in each test cage. The gies). Sense and antisense oligonucleotide primers were designed using subjects were allowed to explore the test cages for 15 min and then PrimerExpress software (Applied Biosystems). The sequences of the removed. The whole procedure took place during the first half hour of primers were as follows: Ghrh sense: 5⬘-GCCATCTTCACCACCAA-3⬘; the dark period in the behavioral room to profit from the enhanced Ghrh antisense: 5⬘-CCTCCTGCTTGTTCATGATGT-3⬘; Sst sense: 5⬘-TC activity of the animals in that period. The papers were then removed TGCATCGTCCTGGCTTT-3⬘; Sst antisense: 5⬘-CTTGGCCAGTTCCT from the test cages and photographed under UV light (UVP GTTTCC-3⬘; cyclophilin sense: 5⬘-GTGGCAAGATCGAAGTGGAGAA White/UV Transilluminator). Finally, the number of urine spots de- AC-3⬘; cyclophilin antisense 5⬘-TAAAAATCAGGCCTGTGGAATGTG- posited on each paper was counted manually, assisted by Photoshop 3⬘. Semiquantitative determination of Ghrh, Sst, and cyclophilin cDNA 7.0. Spots smaller than 1 mm or dragging marks were not considered were performed by kinetic PCR using 9.4 l of TAQurate GREEN Real- in the analysis.
Time PCR MasterMix (Epicenter Biotechnologies) containing 100 ng of Statistics. One- or two-way ANOVA, repeated-measures ANOVA, cDNA, 0.4 M primers, 10 mM Tris-HCl, 50 mM KCl, 3 mM MgCl , 0.2 ANOVA on ranks (Kruskal-Wallis on ranks), Student's test, one-tailed M deoxy-NTP, and 1.25 U Taq polymerase in a final volume of 10 l.
analysis, and 2 2 ⫻ 2 contingency tables were applied as appropriate.
After denaturation at 95°C for 10 min, the cDNA products were ampli- Fisher LSD or Dunnett's contrasts were analyzed post hoc. In all experi- fied with 40 cycles, each cycle consisting of denaturation at 95°C for 30 s, ments, data are represented as means ⫾ SEM.
annealing at 61°C for 1 min, and extension at 72°C for 30 s. The accumu-lating DNA products were monitored by the ABI 7500 sequence detec- tion system (Applied Biosystems) and data were stored continuously Mice lacking D2Rs in pituitary lactotropes display normal
during the reaction. The calculations of the initial mRNA copy numbersin each sample were made according to the cycle threshold (CT) method body growth curves
as described previously The relative Ghrh To inactivate Drd2 expression in pituitary lactotropes, we first or Sst gene expression was normalized to that of the cyclophilin house- generated transgenic mice carrying 2.9 kb of 5⬘ flanking se- keeping gene using the standard curve method. Results are expressed as quences of the mouse prolactin gene ligated upstream of nuclear- arbitrary units (AU) for comparison among samples. AU was defined as targeted cre recombinase coding sequences. A selected pedigree, the expression level relative to a sample of wild-type mice (calibrator Tg(Prl-cre) 1Mrub, expressed functional cre in most PRL- producing cells of the anterior pituitary in a highly selective Food intake. Determination of food intake was performed in isolated manner as determined by immunofluorescence performed on adult Drd2 loxP/loxP or neuroDrd2KO mice. Animals and food (regular coronal pituitary sections of double transgenic mice obtained by chow) were weighed daily at the same hour during the light cycle (15:00 crossing Tg(Prl-cre) 1Mrub mice with the cre reporter mouse line Ai14 that expresses the fluorescent protein Behavioral testing. Haloperidol-induced catatonia was determined us- ing the horizontal bar test in which the time of immobility after 30 min of td-tomato upon cre recombination ). LacDrd2KO mice a single injection of saline or 1.5 mg/kg haloperidol (Tocris Bioscience) were obtained by successively breeding Tg(Prl-cre) 1Mrub mice was assessed. Five trials were performed for each animal with a 180 s with Drd2 loxP/loxP mice In situ hybridization cutoff for each trial. For the analysis, we considered the maximum time of analysis using a Drd2 exon 2 antisense riboprobe showed that
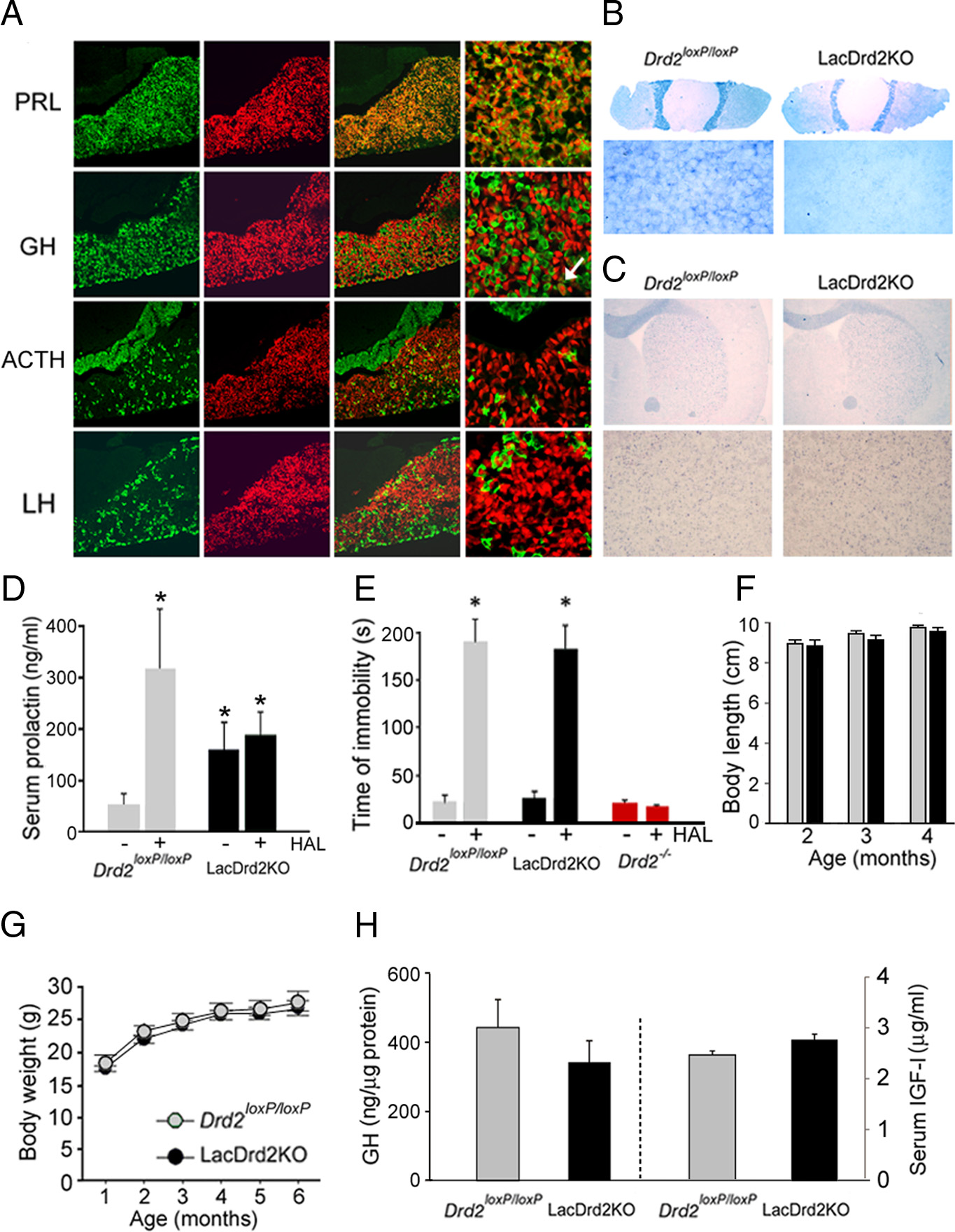
Noaín, Pe´rez-Milla´n et al. • Central D2Rs in Body Growth and Social Dominance
J. Neurosci., March 27, 2013 • 33(13):5834 –5842 • 5837
lacDrd2KO mice of both genders, in con-trast to the effect observed in Drd2loxP/loxPcontrol mice, demonstrating the total ab-sence of functional D2Rs in lactotropes ofconditional lacDrd2KO mice ). Wealso found that haloperidol was equallyeffective
lacDrd2KO and Drd2loxP/loxP control mice,in contrast to Drd2⫺/⫺ mice, which wereinsensitive to the D2R blocker's effect ), indicating that central D2R function isintact in lacDrd2KO mice. Therefore,lacDrd2KO mice are authentic lactotropeD2R-deficient mice. Body size and bodyweight curves of lacDrd2KO male micewere normal and identical to those observedin Drd2loxP/loxP male siblings ,G), aswell as GH axis parameters such as pituitaryGH and serum IGF-I levels, which were alsonormal in lacDrd2KO males ).
These results indicate that the absence ofD2Rs in pituitary lactotropes is not likely toaccount for the dwarfism found in Drd2⫺/⫺male mice.
NeuroDrd2KO mice have reduced body
weight and size
Transgenic mice expressing cre from a rat
nestin promoter were used in
combination with Drd2 loxP/loxP mice to
inactivate Drd2 alleles in neural progeni-
tors. As a result, neuroDrd2KO mice carry
null Drd2 alleles in cells of neural origin
and normally express Drd2 in other cell
types, as demonstrated at the molecular
and functional levels In situ
hybridization assays using a Drd2 exon 2
antisense riboprobe ) and [ 3H]-
nemonapride binding autoradiography
Figure 1.
LacDrd2KO mice display hyperprolactinemia and normal body growth. A, Cre-mediated td-tomato expression in
) revealed the absence of Drd2
various pituitary cell types. Immunofluorescence using primary antisera for PRL, GH, ACTH, and LH (green) performed on coronal
mRNA and D2Rs, respectively, on coronal
pituitary sections of Tg(Prl-cre) 1Mrub mice crossed with the cre inducible td-tomato line Ai14 (first column). Identical sections show
brain sections taken from neuroDrd2KO
td-tomato fluorescence (red, second column). Superimposed images show coexpression of cre and PRL but not GH (with a few
exceptions, see white arrow), ACTH or LH at lower (third column) and higher magnification (forth column). B, In situ hybridization
mice. NeuroDrd2KO mice failed to show
using a Drd2 exon 2 antisense riboprobe in pituitary and brain sections of lacDrd2KO. Shown are whole pituitary sections and higher
haloperidol-induced catatonia, in con-
magnification of the anterior lobe and coronal brain sections (C) at the level of striatum and higher magnification. D, Serum PRL
trast to Drd2 loxP/loxP mice, but similar to
levels in Drd2 loxP/loxP (n ⫽ 10) and lacDrd2KO (n ⫽ 11) females receiving saline (-) or haloperidol 3 mg/kg intraperitoneally (HAL,
Drd2 ⫺/⫺ mice ), indicating the
⫹). Two-way ANOVA *p ⬍ 0.01 versus saline-treated Drd2loxP/loxP mice. E, Haloperidol (1.5 mg/kg, i.p.)-induced catatonia.
absence of functional central D2Rs. In ad-
Latency to remove the front paws from an elevated horizontal bar was evaluated in Drd2 loxP/loxP (n ⫽ 4), lacDrd2KO (n ⫽ 5), and
dition, D2R levels in the pituitary glands
Drd2 ⫺/⫺ (n ⫽ 3) male mice. *p ⬍ 0.01 versus saline for each genotype. F, Nose to anus length of neuroDrd2KO and Drd2 loxP/loxP
of neuroDrd2KO mice appeared to be
male littermates. G, Body weight curves of Drd2 loxP/loxP and lacDrd2KO male littermates. No significant differences were found
similar to those observed in Drd2 loxP/loxP
between genotypes. H, Radioimmunoassay showed normal pituitary GH and serum IGF-I levels in lacDrd2KO mice relative to their
mice , B) and functionally nor-
Drd2 loxP/loxP littermates. Bars and circles represent the mean ⫾ SEM.
mal, as evidenced by the normal prolactinlevels observed in neuroDrd2KO mice
lacDrd2KO mice lacked D2Rs in pituitary lactotropes while re-
) and the increase observed after receiving a 3 mg/kg
taining normal Drd2 expression in pituitary melanotropes of the
intraperitoneal haloperidol injection ). Therefore,
intermediate lobe ) and in all brain areas examined
neuroDrd2KO mice lack D2Rs in the CNS but retain normally
). To investigate whether ablation of D2R in lacDrd2KO mice
functional D2Rs in the anterior pituitary. NeuroDrd2KO male
was complete, we measured basal serum PRL levels and, more
mice showed reduced body weight ) and length )
importantly, the acute effect of the D2R blocker haloperidol.
compared with their Drd2loxP/loxP male littermates. Body weight of
LacDrd2KO mice displayed hyperprolactinemia that was par-
neuroDrd2KO mice was 14.2% lower than Drd2loxP/loxP at 2 months
ticularly significant in females ). The D2R antagonist halo-
of age and this difference increased over time, being 21.7% lower at 6
peridol (3 mg/kg, i.p.) failed to increase serum PRL levels in
months ). Body size of neuroDrd2KO mice at weaning was
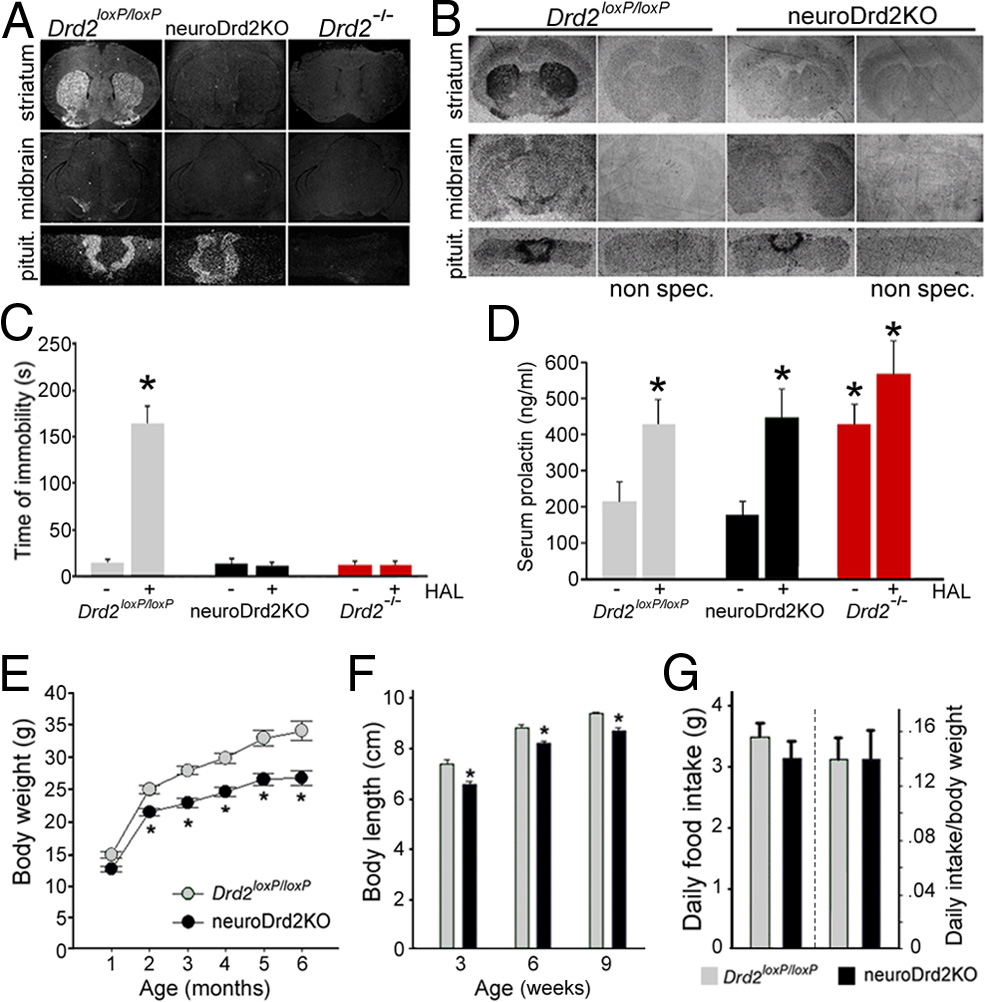
5838 • J. Neurosci., March 27, 2013 • 33(13):5834 –5842
Noaín, Pe´rez-Milla´n et al. • Central D2Rs in Body Growth and Social Dominance
Figure 2.
NeuroDrd2KO mice have reduced body weight and size. A, B, In situ hybridization
assay using a Drd2 exon 2 [ 35S]-UTP antisense riboprobe (A) and [ 3H]-nemonapride binding
autoradiography (B) in brain and pituitary coronal sections of Drd2 loxP/loxP and neuroDrd2KO
mice. Sections of Drd2 ⫺/⫺ mice were included as negative controls. C, Haloperidol (HAL, 1.5
mg/kg, i.p.)-induced catatonia. Latency to remove the front paws from an elevated horizontal
bar was evaluated in neuroDrd2KO mice and compared with Drd2 loxP/loxP (n ⫽ 6) and
Drd2 ⫺/⫺ (n ⫽ 6) mice. *p ⬍ 0.01 haloperidol versus saline for each genotype. D, Serum PRL
Figure 3.
NeuroDrd2KO mice display an impaired GH axis. A, Immunofluorescence using an
levels of female mice (n ⫽ 10 –12) receiving haloperidol (HAL, 3 mg/kg, i.p.) or saline. *p ⬍
anti-hGH primary antiserum performed on 4 m pituitary slices of Drd2 loxP/loxP (n ⫽ 3) and
0.05 haloperidol versus saline for each genotype. E, Body weight curves of neuroDrd2KO and
neuroDrd2KO (n ⫽ 3) mice showed fewer pituitary somatotropes (green) in neuroDrd2KO mice
Drd2 loxP/loxP male mice (n ⫽ 7–12). Repeated-measures ANOVA genotype: F
relative to control mice. Cell nuclei were stained with propidium iodide (red). B, Right, Percent-
p ⬍ 0.005. *p ⬍ 0.01 neuroDrd2KO versus Drd2 loxP/loxP littermates, post hoc Fisher analysis. F,
age of GH-positive cells, one-way ANOVA (OWA): F
⫽ 18.01, *p ⬍ 0.02. Left, radioimmu-
Nose to anus length of neuroDrd2KO and Drd2 loxP/loxP male mice (n ⫽ 9 –11) measured 3, 6,
noassay of GH concentration in anterior pituitary glands of Drd2 loxP/loxP (n ⫽ 7) and
and 9 weeks after birth. Repeated-measures ANOVA genotype: F
⫽ 21.35, p ⬍ 0.005.
neuroDrd2KO (n ⫽ 10) siblings (OWA: F
⫽ 7.54, *p ⬍ 0.05). C, Right, relative hypotha-
*p ⬍ 0.05 for neuroDrd2KO versus age-matched Drd2 loxP/loxP littermates, post hoc Fisher anal-
lamic mRNA levels of Ghrh in Drd2 loxP/loxP (n ⫽ 9) and neuroDrd2KO (n ⫽ 9) mice determined
ysis. G, Food consumption was measured for 7 consecutive days in adult neuroDrd2KO and
by semiquantitative real-time PCR (OWA: F
⫽ 6.32, *p ⬍ 0.05). Left, relative hypotha-
Drd2 loxP/loxP (n ⫽ 5–10) mice. Averaged food intake (left) and food intake relative to animal
lamic mRNA levels of Sst from Drd2 loxP/loxP and neuroDrd2KO mice (n ⫽ 13). Samples were
weight (right) is plotted. Bars and circles represent the mean ⫾ SEM.
normalized to cyclophilin mRNA. Bars denote the mean ⫾ SEM. All mice were 6 –9 months old.
the nervous system develop an immature GH axis that limits the
already significantly lower (10.2%) than control littermates and the
body growth program.
difference persisted until reaching adulthood ). The de-creased growth exhibited by neuroDrd2KO male mice was not
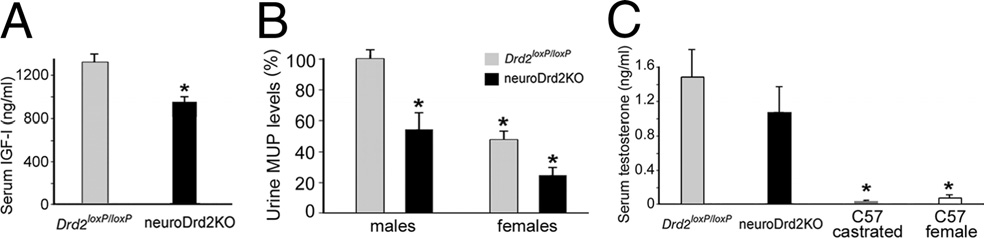
NeuroDrd2KO mice have low levels of the GH-dependent
due to reduced feeding, because the daily food intakes of
liver proteins IGF-I and MUP
neuroDrd2KO and Drd2loxP/loxP siblings were similar ).
To determine whether the impaired GH axis found in
Therefore, these results demonstrate that the sole absence of D2Rs in
neuroDrd2KO mice has downstream consequences in GH-
the nervous system impairs normal growth rates and produces
dependent liver hormones, we measured IGF-I and MUP levels.
smaller mice.
Serum IGF-I levels were 18.7% lower in neuroDrd2KO malemice compared with those found in their control siblings
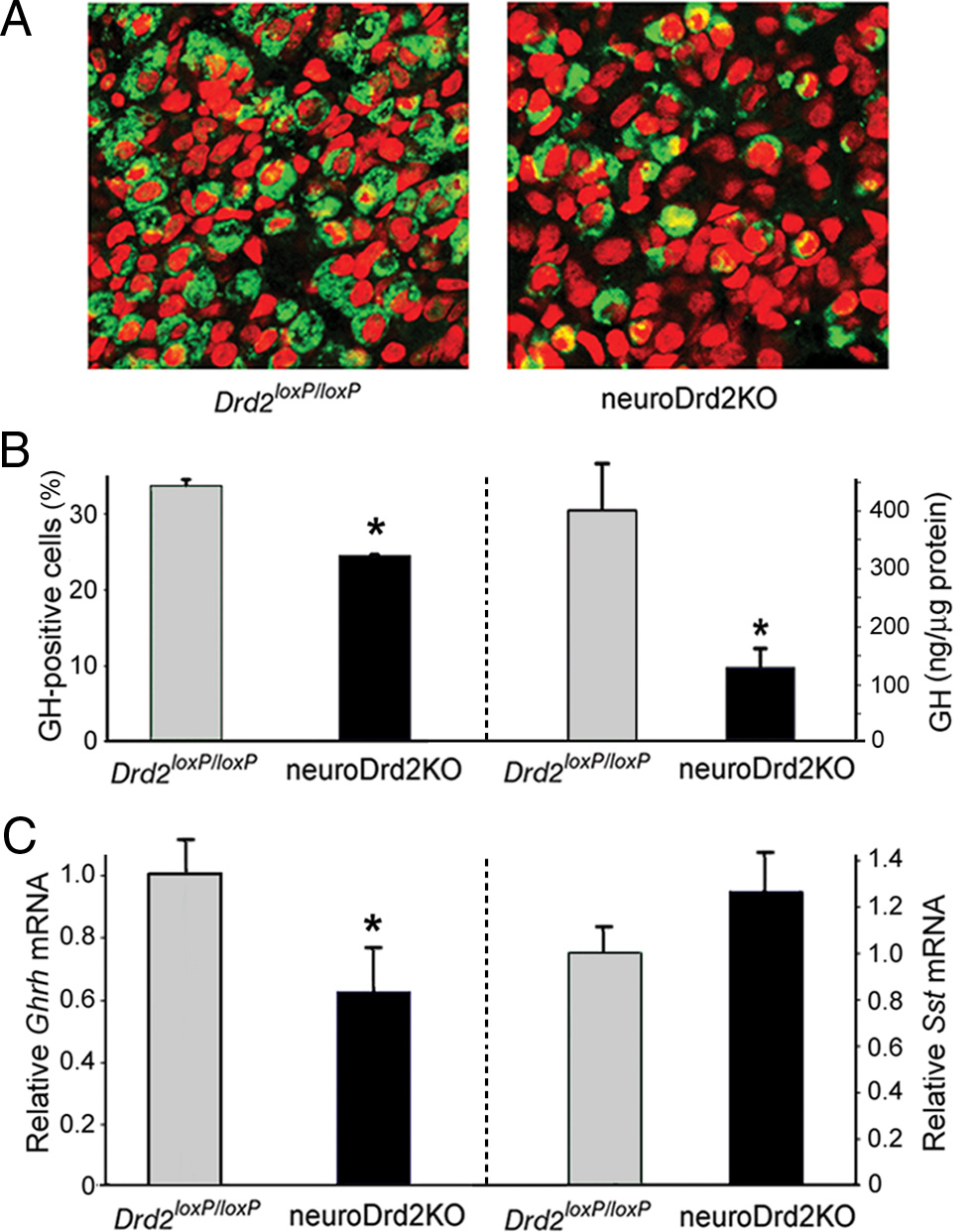
NeuroDrd2KO mice have an impaired GH axis
), whereas MUP levels were markedly reduced (45.6%) in the
We analyzed the relative number of somatotropes and GH con-
urine of neuroDrd2KO male mice ). Interestingly, MUP
tent in pituitaries from neuroDrd2KO and control male mice.
levels of the male mutants were similar to those found in the urine
Immunofluorescence performed on pituitary slices taken from
of their Drd2 loxP/loxP female littermates ). In contrast,
neuroDrd2KO male mice showed a 28.1% shortfall of GH-
serum testosterone levels in neuroDrd2KO male mice were sim-
positive cells compared with control male mice , B). In
ilar to those found in control male littermates, in clear contrast to
agreement, a great reduction (63.2%) in GH content was deter-
testosterone levels measured in adult wild-type females and cas-
mined by radioimmunoassay in pituitaries from neuroDrd2KO
trated wild-type males ). These results suggest that the
mice ). Analysis of the hypothalamic content of Ghrh and
reduced MUP levels were primarily the consequence of an im-
Sst mRNAs using a semiquantitative RT-PCR assay showed that
paired GH axis.
neuroDrd2KO males had 30% lower Ghrh mRNA levels com-pared with their Drd2 loxP/loxP littermates ), whereas hy-
Central D2Rs, social dominance, and territorial behavior
pothalamic Sst mRNA levels were not significantly altered
The higher MUP content typically found in the urine of adult
). These results demonstrate that male mice lacking D2Rs in
males is thought to promote a variety of male-specific behaviors
Noaín, Pe´rez-Milla´n et al. • Central D2Rs in Body Growth and Social Dominance
J. Neurosci., March 27, 2013 • 33(13):5834 –5842 • 5839
Figure 4.
NeuroDrd2KOmice have reduced IGF-I and MUPs and normal testostene levels. A,
Radioimmunoassay of serum IGF-I levels in neuroDrd2KO (n ⫽ 14) and Drd2 loxP/loxP (n ⫽ 20)mice (one-way ANOVA [OWA]: F
from male Drd2 loxP/loxP (n ⫽ 17) and neuroDrd2KO (n ⫽ 12) adult mice and from theirDrd2 loxP/loxP (n ⫽ 16) and neuroDrd2KO (n ⫽ 9) female littermates. Two-way ANOVA showedsignificant main effects both for sex and genotype (sex effect: F
⫽ 26.68, *p ⬍ 0.00001
and genotype effect: (1,50) ⫽ 17.84, *p ⬍ 0.0005). C, Serum testosterone levels determined
by radioimmunoassay in neuroDrd2KO male mice (n ⫽ 9) compared with their Drd2 loxP/loxP
male littermates (n ⫽ 9). Castrated wild-type adult male mice (n ⫽ 4) and wild-type adult
females (n ⫽ 5) were included as negative controls (OWA: F
⫽ 5.0673, p ⬍ 0.008). *p ⬍
0.05 for wild-type castrated males and wild-type females versus Drd2 loxP/loxP and neuroDrd2KOmale mice, post hoc Fisher's LSD. Bars represent the mean ⫾ SEM.
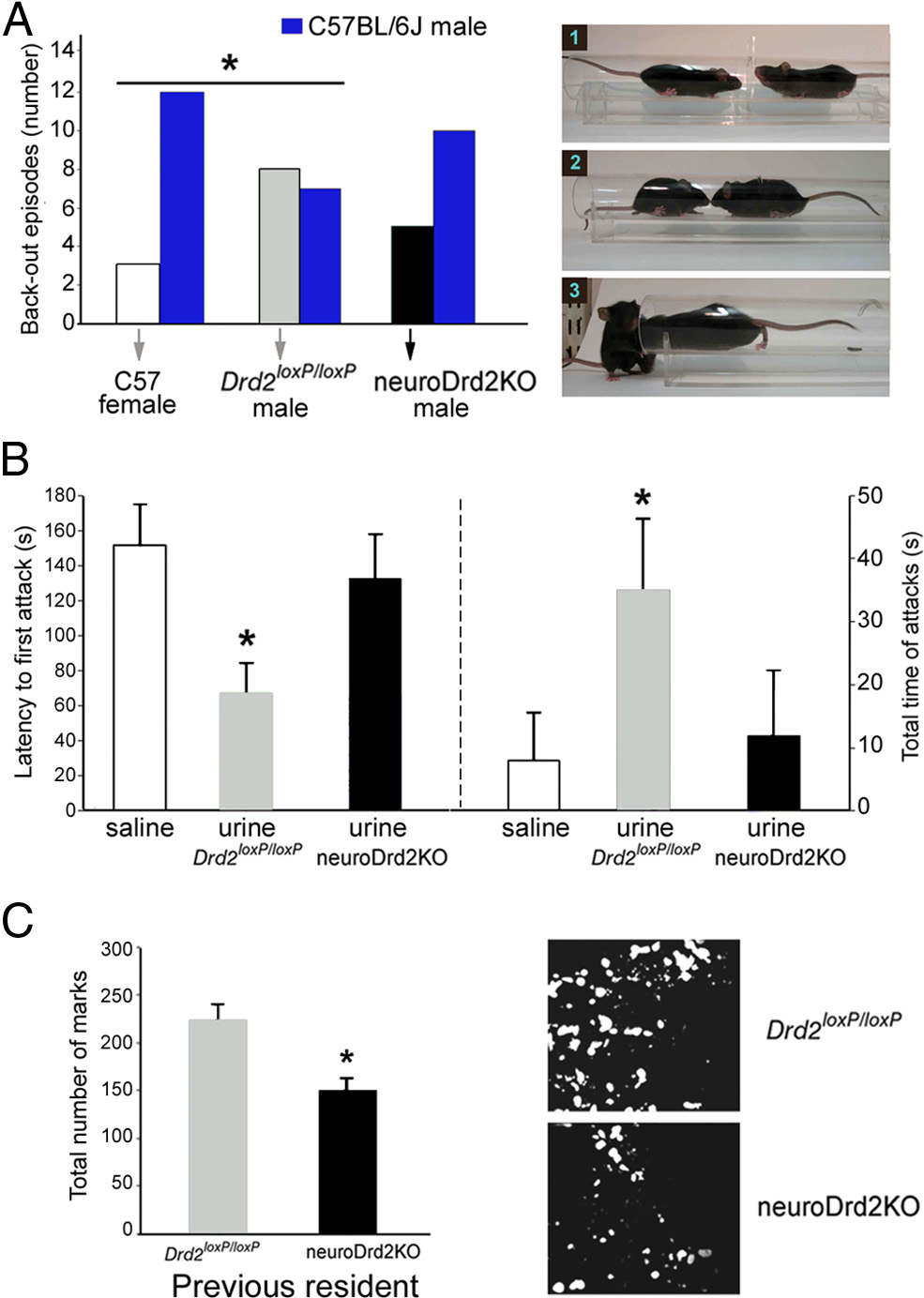
that are critical for reproductive fitness, such as social recogni-tion, territorial scent-marking, and aggression To evaluate whether the feminized lowerMUP levels found in the urine of neuroDrd2KO male mice mod-ify these evolutionarily conserved behaviors, we designed a seriesof behavioral tests. First, we examined how adult male C57BL/6JWT mice reacted when confronted with either Drd2 loxP/loxP orneuroDrd2KO male adults in a social dominance experimentalparadigm called the double entrance tube test Normally in this test, when two adult male mice are placed at theopposite extremes of a cylindrical tube, they both gain access tothe center and, after close mutual inspection, one moves forward,forcing the other to walk backwards until stepping down from the
Figure 5.
Urine from adult neuroDrd2KO males fails to promote social and territorial behav-
end of the tube , right). However, when confronting fe-
iors. A, Left, C57BL/6J adult females, Drd2 loxP/loxP adult males and neuroDrd2KO adult males
males, adult male mice do not show this behavior and back out
were challenged with C57BL/6J adult males in a social-dominance tube test. Fifteen trials were
spontaneously without showing signs of social dominance. When
performed for each genotype. The behavior of C57BL/6J males was significantly different when
we challenged C57BL/6J adult males with sex- and age-matched
confronting C57BL/6J females compared with Drd2 loxP/loxP males (one-tailed 2 *p ⬍ 0.05).
Drd2 loxP/loxP mice, we observed a similar number of retreats in
C57BL/6J males confronting neuroDrd2KO males showed an intermediate phenotype because
mice of each genotype: C57BL/6J males backed out from the tube
their behavior was not different from that observed when confronting either C57BL/6J females
in seven of 15 trials, whereas Drd2 loxP/loxP males backed out in the
or Drd2 loxP/loxP males (one-tailed 2 p ⬎ 0.05). Right, Representative images of a typical test:(1) two mice approaching the center of the tube, (2) mutual inspection, and (3) the mouse on
other eight ). Conversely, when challenged with WT fe-
the right pushes the other out. B, Intruder-resident test. Isolated C57BL/6J adult male residents
males, C57BL/6J adult males backed out of the tube in 12 of 15
(n ⫽ 18) were challenged with C57BL/6J castrated males swabbed on their backs and genital
encounters without showing social dominance behavior
areas with saline (n ⫽ 6), urine from Drd2 loxP/loxP adult male mice (n ⫽ 6), or urine from
). Interestingly, C57BL/6J adult male mice challenged against
neuroDrd2KO adult males (n ⫽ 6). Left, latency to the first resident's attack (one-way ANOVA
sex- and age-matched neuroDrd2KO mice exhibited behavioral
⫽ 4.55, p ⬍ 0.05). *p ⬍ 0.05 compared with saline, post hoc Fisher analysis.
responses similar to those shown when challenged against
Right, total time spent attacking (Kruskal-Wallis OWA on ranks: H ⫽ 6.65 with 2 degrees of
C57BL/6J females. In this case, C57BL/6J males backed out of the
freedom, p ⬍ 0.05). *p ⬍ 0.05 compared with saline, post hoc Dunnett's comparison. C, Scent
tube spontaneously in 10 of 15 encounters and moved forward
marking test. Left, C57BL/6J adult male mice were challenged in a cage previously occupied by
neuroDrd2KO males in only five trials ). The different
a Drd2flox/flox (n ⫽ 5) or a neuroDrd2KO (n ⫽ 8) adult male mouse. Urine marks made by the
reaction of C57BL/6J adult males toward neuroDrd2KO adult
visitor during 5 min were counted under UV light (OWA: F
14.66, *p ⬍ 0.005). Right,
two representative examples of spotted urine are shown. Bars represent the mean ⫾ SEM.
males compared with that observed when confronting theirDrd2 loxP/loxP littermates could be due, at least in part, to the rel-atively smaller size of neuroDrd2KO mice and/or to their lower
of the intruder, resident mice exhibited aggressive behavior that
concentration of urinary MUP. To further investigate this lat-
varied in latency and intensity depending on the nature of the
ter hypothesis, we tested the ability of the urine taken from
liquid swabbed on the intruder. Latency to the first attack was
Drd2 loxP/loxP and neuroDrd2KO male adults to promote territo-
65 ⫾ 17 s when castrated mice received urine from Drd2loxP/loxP
rial male-to-male aggression in an adapted version of the
mice, and this value increased significantly when mice were
intruder-resident aggression test To
swabbed with either saline or urine taken from neuroDrd2KO
this end, each C57BL/6J adult male resident mouse reared in
male adult mice , left). Moreover, 3 of 6 intruders
isolation for 1 month was challenged with a castrated adult WT
swabbed with saline and 3 of 6 intruders swabbed with
male intruder mouse placed into the resident's cage. Each cas-
neuroDrd2KO male mice urine received no attacks from the res-
trated mouse was swabbed on its back and genital area with
ident males during the 3 min test. Therefore, when resident males
saline or freshly collected urine taken from Drd2 loxP/loxP or
were confronted with castrated males swabbed with urine taken
neuroDrd2KO adult male mice. Upon close olfactory inspection
from neuroDrd2KO mice, the latency to the first attack increased
5840 • J. Neurosci., March 27, 2013 • 33(13):5834 –5842
Noaín, Pe´rez-Milla´n et al. • Central D2Rs in Body Growth and Social Dominance
markedly and the time spent attacking decreased compared with
neurons of the indirect extrapyramidal pathway
castrated intruders swabbed with urine taken from Drd2 loxP/loxP
because these two conditional Drd2 knock-out models
male adults , right). Finally, we performed a third exper-
exhibited normal growth curves. Because D2Rs couple to inhib-
iment to investigate whether C57BL/6J adult male mice displayed
itory signals that diminish neuron activity, a candidate neuronal
different scent-marking behavior when placed in a cage where a
type is a group of periventricular SST neurons that make synaptic
sex- and age-matched Drd2 loxP/loxP or a neuroDrd2KO mouse
contacts with GHRH arcuate neurons to exert a tonic inhibition
was previously housed. A stereotyped behavior commonly ob-
of Ghrh expression and GHRH release
served when a male mouse detects odorant cues from another
and control the rhyth-
adult conspecific male is the induction of widespread urine de-
mic oscillations of GHRH neuronal firing that establish the male
positions that are interpreted as increased scent-marking activity
pattern of GH release
to cover the area with its own scent This
However, a semiquantitative RT-PCR determination only
behavior is greatly reduced when the adult male is placed in a
found a tendency for increased hypothalamic Sst mRNA levels
location previously visited only by a castrated or a subordinate
in neuroDrd2KO mice that never reached statistical signifi-
male By measuring the number of
urinary spots, we determined that C57BL/6J adult male mice
The present study demonstrates that central D2Rs regulate a
reared in isolation displayed greater scent-marking activity when
neuroendocrine/exocrine cascade that controls body growth and
placed in a cage previously occupied by a Drd2 loxP/loxP mouse
participates in male-to-male territorial behavior. This multistep
rather than by a neuroDrd2KO male adult littermate ).
signaling cascade initiates in brain centers that regulate the hypo-
These results indicate that wild-type male adults do not exhibit
thalamic level Ghrh expression and GHRH release, which then
typical male-to-male social and territorial behavior when ex-
relays to the pituitary to control the pattern of GH release. GH
posed to neuroDrd2KO males or to their urine. Therefore, cen-
surges from the pituitary of male rodents are characterized by
tral D2Rs are necessary for the masculinization of the GH axis
high-amplitude and low-frequency pulses intercalated among
and, therefore, for the production of higher levels of pheromones
long nadir periods, which permit the resensitization of GH recep-
typically found in the urine of normal males.
tor signaling by a Janus kinase/signal transducer and activation oftranscription (JAK-STAT) complex that
specifically involves STAT5a and STAT5b
In this study, we conducted a differential cell-specific ablation
Only when long interpulse
strategy to eliminate D2Rs selectively from pituitary lactotropes
nadirs of GH levels are attained in puberal males are high tran-
and, independently, from brain neurons. Whereas lacDrd2KO
scriptional rates of Igf1 and other somatomedins that promote
mice displayed normal body growth, neuroDrd2KO mice
body growth observed in the liver and extrahepatic sites
showed deficits in the GH axis that led to reduced body weight
The lower serum levels of IGF-I observed in
and length, recapitulating the dwarf phenotype observed in
neuroDrd2KO mice are consistent with their smaller body size
Drd2 ⫺/⫺ mutants. Based on the analysis of lacDrd2KO mice, we
and weight, a phenotype that in the wild could be detrimental for
concluded that the absence of lactotrope D2R is not responsible
reproductive fitness due to reduced competitiveness among
for the altered lactotrope/somatotrope ratio previously observed
males. The sexually dimorphic liver proteins that depend on the
in Drd2 ⫺/⫺ mice, ruling out its participation in the termi-
male pattern of GH release is manifold and include MUPs
nal differentiation of these two cell types. Furthermore,
This group of pheromones se-
neuroDrd2KO mice showed fewer somatotropes, diminished pi-
creted into the blood and then excreted in the urine are known to
tuitary GH content, and lower levels of hypothalamic Ghrh ex-
promote social recognition and territorial dominance between
pression. Therefore, central D2Rs are essential for the normal
males Therefore, an
maturation of the GH axis and full development of the body
emerging downstream finding disclosed by this study is that the
growth plan. Selective ablation of D2Rs in the brain while main-
urine of mice lacking D2Rs in the nervous system showed a lower
taining normal Drd2 expression in the pituitary was achieved by
content of MUP and, therefore, lost the signaling cues that trigger
using a strain of transgenic mice expressing cre from a nestin
territorial behavior in other males. Typically, MUP levels in males
promoter. A similar strategy has been used previously to elimi-
are much higher than in females and this
nate the transcription factor CREB from the CNS
difference not only depends on the sexually dimorphic pattern
because the nestin gene (Nes) is expressed in
of GH pulsatility, but also on testosterone concentration, be-
embryonic dividing cells derived from the neuroectoderm but
cause castrated males have feminized MUP levels and females
not from the oral ectoderm
treated with testosterone display male MUP levels
and is particularly absent from progenitors and differ-
Because testosterone concentration was normal in
entiated cells of all anterior pituitary cell lineages
neuroDrd2KO mice, the reduced MUP levels are likely to be the
In fact, neuroDrd2KO mice
sole consequence of an impaired GH axis resulting from lack of
showed normal Drd2 expression in the intermediate and anterior
Drd2 expression in the CNS. MUPs are encoded by ⬎30 genes,
lobe of the pituitary but complete absence of expression in the
mainly synthetic, in a locus of mouse chromosome 4
CNS. The differential response to haloperidol-induced hyperp-
The large allelic variation at this
rolactinemia and catatonia observed in lacDrd2KO and
locus and the relative amount of each MUP in the mouse urine
neuroDrd2KO mice confirmed, at the functional level, our suc-
contributes to generate a great combinatorial diversity of individ-
cessful attempt to inactivate Drd2 expression selectively in the
ual patterns that are believed to provide, in the wild, a unique
pituitary or in the brain.
biological identity card for each conspecific
Identifying the Drd2-expressing neurons in the CNS control-
In fact, male mice use MUPs to scent-mark and counter-
ling the maturation of the GH axis is still pending, although our
mark territories The ability of MUPs
own studies ruled out the participation of DA neurons expressing
to create a chemical barrier for territorial defense and to promote
D2 autoreceptors and striatal medium spiny
territorial protection adds to the adaptive behavioral inventory
Noaín, Pe´rez-Milla´n et al. • Central D2Rs in Body Growth and Social Dominance
J. Neurosci., March 27, 2013 • 33(13):5834 –5842 • 5841
that males use to gain access to receptive females. Therefore, this
García-Tornadu I, Díaz-Torga G, Risso GS, Silveyra P, Cataldi N, Ramirez
study shows that central D2R regulate a neuroendocrine-
MC, Low MJ, Libertun C, Becu´-Villalobos D (2009) Hypothalamic
exocrine cascade that controls the maturation of the GH axis and
orexin, OX1, alphaMSH, NPY and MCRs expression in dopaminergicD2R knockout mice. Neuropeptides 43:267–274.
downstream signals that play important roles in territorial behav-
Gleiberman AS, Michurina T, Encinas JM, Roig JL, Krasnov P, Balordi F,
ior and competition between adult males.
Fishell G, Rosenfeld MG, Enikolopov G (2008) Genetic approaches
Throughout vertebrate evolution, the D2R has played key
identify adult pituitary stem cells. Proc Natl Acad Sci U S A 105:6332–
roles in the complex repertoire of adaptive functions that im-
prove fitness, reproductive success, and survival. For example,
Godfrey P, Rahal JO, Beamer WG, Copeland NG, Jenkins NA, Mayo KE
D2Rs in the nucleus accumbens and striatum participate in neu-
(1993) GHRH receptor of little mice contains a missense mutation in
ral circuits that assign reinforcing and hedonic value to nutritious
the extracellular domain that disrupts receptor function. Nat Genet4:227–232.
food and reproductive sex
Hastie ND, Held WA, Toole JJ (1979) Multiple genes coding for the
Moreover, this system ac-
androgen-regulated major urinary proteins of the mouse. Cell 17:
quired the exquisite capacity to anticipate and predict the close
availability of food and sexual mates by providing incentive sa-
Hurst JL, Beynon RJ (2004) Scent wars: the chemobiology of competitive
lience to conditioned environmental stimuli
signalling in mice. BioEssays 26:1288 –1298.
D2Rs are fundamental also in
Hurst JL, Payne CE, Nevison CM, Marie AD, Humphries RE, Robertson DH,
motor coordination, locomotor activity, and executive planning,
Cavaggioni A, Beynon RJ (2001) Individual recognition in mice medi-ated by major urinary proteins. Nature 414:631– 634.
all functions that allow animals to expand the territories used to
Kaplan AR, Doyle T, Casey E, Free RB, Sibley DR, Rubenstein M, Alvarez VA
search for natural rewards. In addition, it has been shown that
(2012) Distinct roles of dopamine D2 receptors in dorsal and ventral
central D2Rs participate in establishing the social dominance
striatum on motor and drug-related behaviors. Paper presented at the
ranking within males In mammalian fe-
Society for Neuroscience Annual Meeting, New Orleans, LA, 2012.
males, D2Rs of pituitary lactotropes mediate a functional switch
Kelley AE, Berridge KC (2002) The neuroscience of natural rewards: rele-
that is essential for reproduction: although tuberoinfundibular
vance to addictive drugs. J Neurosci 22:3306 –3311.
dopamine normally inhibits PRL release to ensure fertility, D2R
Kelly MA, Rubinstein M, Asa SL, Zhang G, Saez C, Bunzow JR, Allen RG,
Hnasko R, Ben-Jonathan N, Grandy DK, Low MJ (1997) Pituitary lac-
stimulation is suppressed before labor to greatly increase circu-
totroph hyperplasia and chronic hyperprolactinemia in dopamine D2
lating PRL levels that exert lactogenic and social bonding prop-
receptor-deficient mice. Neuron 19:103–113.
erties that are necessary to raise the newborns efficiently. In
Krylyshkina O, Chen J, Mebis L, Denef C, Vankelecom H (2005) Nestin-
summary, our study reports two additional adaptive properties of
immunoreactive cells in rat pituitary are neither hormonal nor typical
central D2Rs: (1) high levels of somatotropic hormones allow
folliculo-stellate cells. Endocrinology 146:2376 –2387.
adult male mice to fulfill their genetic body growth program and
Lijam N, Paylor R, McDonald MP, Crawley JN, Deng CX, Herrup K, Stevens
reach a fit body size and, in parallel, (2) adult males excreting
KE, Maccaferri G, McBain CJ, Sussman DJ, Wynshaw-Boris A (1997)Social interaction and sensorimotor gating abnormalities in mice lacking
large amounts of MUPs in their urine are able to protect their
Dvl1. Cell 90:895–905.
territories by scent-marking an area that potential intruders will
Lin SC, Lin CR, Gukovsky I, Lusis AJ, Sawchenko PE, Rosenfeld MG (1993)
recognize as being defended by a fully developed active male.
Molecular basis of the little mouse phenotype and implications for celltype-specific growth. Nature 364:208 –213.
Liposits Z, Merchenthaler I, Paull WK, Flerko´ B (1988) Synaptic communi-
Alba M, Salvatori R (2004) A mouse with targeted ablation of the growth
cation between somatostatinergic axons and growth hormone-releasing
hormone-releasing hormone gene: a new model of isolated growth hor-
factor (GRF) synthesizing neurons in the arcuate nucleus of the rat. His-
mone deficiency. Endocrinology 145:4134 – 4143.
tochemistry 89:247–252.
Asa SL, Kelly MA, Grandy DK, Low MJ (1999) Pituitary lactotroph adeno-
Low MJ, Otero-Corchon V, Parlow AF, Ramirez JL, Kumar U, Patel YC,
mas develop after prolonged lactotroph hyperplasia in dopamine D2
Rubinstein M (2001) Somatostatin is required for masculinization of
receptor-deficient mice. Endocrinology 140:5348 –5355.
growth hormone-regulated hepatic gene expression but not of somatic
growth. J Clin Invest 107:1571–1580.
Bello EP, Mateo Y, Gelman DM, Noaín D, Shin JH, Low MJ, Alvarez VA,
Madisen L, Zwingman TA, Sunkin SM, Oh SW, Zariwala HA, Gu H, Ng LL,
Lovinger DM, Rubinstein M (2011) Cocaine supersensitivity and en-
Palmiter RD, Hawrylycz MJ, Jones AR, Lein ES, Zeng H (2010) A robust
hanced motivation for reward in mice lacking dopamine D2 autorecep-
and high-throughput Cre reporting and characterization system for the
tors. Nat Neurosci 14:1033–1038.
whole mouse brain. Nat Neurosci 13:133–140.
Berridge KC, Robinson TE (1998) What is the role of dopamine in reward:
Mantamadiotis T, Lemberger T, Bleckmann SC, Kern H, Kretz O, Martin
hedonic impact, reward learning, or incentive salience? Brain Res Rev
Villalba A, Tronche F, Kellendonk C, Gau D, Kapfhammer J, Otto C,
Schmid W, Schu¨tz G (2002) Disruption of CREB function in brain leads
Chamero P, Marton TF, Logan DW, Flanagan K, Cruz JR, Saghatelian A,
to neurodegeneration. Nat Genet 31:47–54.
Cravatt BF, Stowers L (2007) Identification of protein pheromones that
McCarthy GF, Beaudet A, Tannenbaum GS (1992) Colocalization of so-
promote aggressive behaviour. Nature 450:899 –902.
matostatin receptors and growth hormone-releasing factor immuno-
Cheetham SA, Thom MD, Jury F, Ollier WE, Beynon RJ, Hurst JL (2007)
reactivity in neurons of the rat arcuate nucleus. Neuroendocrinology
The genetic basis of individual-recognition signals in the mouse. Curr
Biol: CB 17:1771–1777.
McIntosh I, Bishop JO (1989) Differential expression in male and female
Davey HW, Park SH, Grattan DR, McLachlan MJ, Waxman DJ (1999)
mouse liver of very similar mRNAs specified by two group 1 major uri-
STAT5b-deficient mice are growth hormone pulse-resistant. Role of
nary protein genes. Mol Cell Biol 9:2202–2207.
STAT5b in sex-specific liver p450 expression. J Biol Chem 274:35331–
Morgan D, Grant KA, Gage HD, Mach RH, Kaplan JR, Prioleau O, Nader SH,
Buchheimer N, Ehrenkaufer RL, Nader MA (2002) Social dominance in
Desjardins C, Maruniak JA, Bronson FH (1973) Social rank in house mice:
monkeys: dopamine D2 receptors and cocaine self-administration. Nat
differentiation revealed by ultraviolet visualization of urinary marking
Neurosci 5:169 –174.
patterns. Science 182:939 –941.
Mu¨ller EE, Locatelli V, Cocchi D (1999) Neuroendocrine control of growth
Díaz-Torga G, Feierstein C, Libertun C, Gelman D, Kelly MA, Low MJ, Ru-
hormone secretion. Physiol Rev 79:511– 607.
binstein M, Becu´-Villalobos D (2002) Disruption of the D2 dopamine
Palmiter RD (2008) Dopamine signaling in the dorsal striatum is essential
receptor alters GH and IGF-I secretion and causes dwarfism in male mice.
for motivated behaviors: lessons from dopamine-deficient mice. Ann
Endocrinology 143:1270 –1279.
New York Acad Sci 1129:35– 46.
5842 • J. Neurosci., March 27, 2013 • 33(13):5834 –5842
Noaín, Pe´rez-Milla´n et al. • Central D2Rs in Body Growth and Social Dominance
Park SH, Liu X, Hennighausen L, Davey HW, Waxman DJ (1999) Distinc-
musculus and Mus musculus domesticus). J Chem Ecol 33:861– 869.
tive roles of STAT5a and STAT5b in sexual dimorphism of hepatic P450
gene expression. Impact of STAT5a gene disruption. J Biol Chem 274:
Tronche F, Kellendonk C, Kretz O, Gass P, Anlag K, Orban PC, Bock R, Klein
R, Schu¨tz G (1999) Disruption of the glucocorticoid receptor gene in the
Porter TE, Hill JB, Wiles CD, Frawley LS (1990) Is the mammosomatotrope
nervous system results in reduced anxiety. Nat Genet 23:99 –103.
a transitional cell for the functional interconversion of growth hormone-
and prolactin-secreting cells? Suggestive evidence from virgin, gestating,
Udy GB, Towers RP, Snell RG, Wilkins RJ, Park SH, Ram PA, Waxman DJ,
and lactating rats. Endocrinology 127:2789 –2794.
Davey HW (1997) Requirement of STAT5b for sexual dimorphism of
Porter TE, Wiles CD, Frawley LS (1991) Evidence for bidirectional in-
body growth rates and liver gene expression. Proc Natl Acad Sci U S A
terconversion of mammotropes and somatotropes: rapid reversion of
94:7239 –7244.
acidophilic cell types to pregestational proportions after weaning. En-
Waxman DJ, Pampori NA, Ram PA, Agrawal AK, Shapiro BH (1991) Inter-
docrinology 129:1215–1220.
pulse interval in circulating growth hormone patterns regulates sexually
Schultz W (2007) Multiple dopamine functions at different time courses.
dimorphic expression of hepatic cytochrome P450. Proc Natl Acad Sci
Annu Rev Neurosci 30:259 –288.
U S A 88:6868 – 6872.
Scully KM, Rosenfeld MG (2002) Pituitary development: regulatory codes
Zimmerman L, Parr B, Lendahl U, Cunningham M, McKay R, Gavin B, Mann
in mammalian organogenesis. Science 295:2231–2235.
J, Vassileva G, McMahon A (1994) Independent regulatory elements in
Stopkova´ R, Stopka P, Janotova´ K, Jedelsky´ PL (2007) Species-specific ex-
the nestin gene direct transgene expression to neural stem cells or muscle
pression of major urinary proteins in the house mice (Mus musculus
precursors. Neuron 12:11–24.
Source: http://ri.conicet.gov.ar/bitstream/handle/11336/2627/Central_Dopamine_D2_Receptors_Regulate_Growth-Hormone____Noa_n_et_al__The_Journal_of_Neuroscience__2013_.pdf?sequence=1&isAllowed=y
osk-ymca-intl.ed.jp
Have a Headache? You Are Not Alone Information about headaches, from the mildly unpleasant to the extremely painful. Transcript of radio broadcast: Vocabulary 25 February 2008 This is SCIENCE IN THE NEWS in VOA Special English. Headache – I'm Barbara Klein. Pain in the head On SCIENCE IN THE NEWS: A guide to headaches Worldwide – Universal;
dktecoenonwovens.in
NONWOVENS BULLETIN Vol. No. 1 Issue No. 05 February 2015 DKTE Centre of Excellence in Nonwovens Plot No. 1, 2 and 3, Shri. Lakshmi Co-Operative Industrial Estate, Hatkanangle – Ichalkaranji ,Dt. Kolhapur – 416109 (MS) India Tel: +91 230 2366354 Email Website: www.dktecoenonwovens.in Nonwoven Bulletin Volume 01 Issue No. 05 / February 2015