Jv129910289p
JOURNAL OF VIROLOGY, Dec. 1999, p. 10289–10295 Copyright 1999, American Society for Microbiology. All Rights Reserved.Effect of Lamivudine on Human T-Cell Leukemia Virus Type 1 (HTLV-1) DNA Copy Number, T-Cell Phenotype, and Anti-Tax Cytotoxic T-Cell Frequency in Patients with HTLV-1-Associated Myelopathy G. P. TAYLOR,1* S. E. HALL,2 S. NAVARRETE,2 C. A. MICHIE,3 R. DAVIS,1 A. D. WITKOVER,2 M. ROSSOR,4 M. A. NOWAK,5† P. RUDGE,6 E. MATUTES,7 C. R. M. BANGHAM,2 AND J. N. WEBER1 Department of Genito-Urinary Medicine and Communicable Diseases, Division of Medicine,1 and Department of Immunology, Division of Investigative Science,2 Imperial College School of Medicine, London W2 1PG, Department of Paediatrics, Ealing Hospital NHS Trust, Ealing, Middlesex UB1 3HW,3 Department of Neurology, St. Mary's Hospital NHS Trust, London W2 1NY,4 Department of Zoology, University of Oxford, Oxford OX1 3PS,5 National Hospital for Neurology and Neurosurgery, London WC1N 3BG,6 and Academic Department of Haematology, Royal Marsden Hospital, London SW3,7 United Kingdom Received 22 March 1999/Accepted 4 September 1999 Patients with human T-cell leukemia virus type 1 (HTLV-1)-associated myelopathy/tropical spastic para-
paresis (HAM/TSP) typically have a high HTLV-1 proviral load in peripheral blood mononuclear cells and
abundant, activated HTLV-1-specific cytotoxic T lymphocytes (CTLs). No effective treatment for HAM/TSP has
been described so far. We report a 10-fold reduction in viral DNA for five patients with HAM/TSP during
treatment with the reverse transcriptase inhibitor lamivudine. In one patient with recent-onset HAM/TSP, the
reduction in viral DNA was associated with a fall in the frequency of CTLs specific to two peptides in the
immunodominant viral antigen Tax. The half-life of peripheral blood mononuclear cell populations was
estimated from changes in viral DNA copy number, CTL frequency, reduction in CD25 expression, and the loss
of dicentric chromosomes following radiation-induced damage. Each of these four different techniques indi-
cated a cellular half-life of approximately 3 days consistent with continuous lymphocyte replication and
destruction. These results indicate that viral replication through reverse transcription significantly contributes
to the maintenance of HTLV-1 viral DNA load. The relative contribution of proliferation versus replication may
vary between infected people.
The human T-cell leukemia virus type 1 (HTLV-1), an en- maintains HTLV-1 proviral load and that the absence of re- demic retrovirus, causes lifelong infection. The majority of verse transcription after a few initial rounds of replication persons infected are asymptomatic carriers. In a minority, might explain the relative conservation of the HTLV-1 genome HTLV-1 infection causes inflammatory diseases characterized compared to the other human retroviruses (32).
by lymphocytic infiltration, of which HTLV-1-associated my- However, a number of observations suggest that viral anti- elopathy/tropical spastic paraparesis (HAM/TSP) is the most gen expression is continuous and that ongoing viral replication notable and causes significant morbidity (7, 20). HTLV-1 also occurs in at least a subpopulation of infected cells. HTLV-1- causes adult T-cell leukemia/lymphoma, an aggressive condi- infected subjects have high titers of antibody to structural gene tion resistant to chemotherapy (9, 30). The proviral load of products throughout the course of the infection (5). HTLV-1 HTLV-1 in peripheral blood mononuclear cells (PBMCs) has mRNA is detected in peripheral blood lymphocytes especially been estimated by diverse techniques: mean proviral load is in patients with a high proviral load (19). A persistent active approximately 10 copies/100 PBMCs in patients with HAM/ cytotoxic T-cell response has been demonstrated for patients TSP and 10-fold less in asymptomatic carriers (16). HTLV-1 with HAM/TSP (11) and for asymptomatic carriers (4). De- proviral load might be maintained either by lymphocyte pro- spite an interisolate diversity of only 4 to 8% between the liferation, with duplication of the HTLV-1 genome at every major HTLV-1 subtypes (12), within-isolate sequence varia- cell division, or by classical retroviral replication via reverse tion and a high dN/dS ratio in the tax gene have been demon- transcription. The relative contribution of these two replica- tion pathways to the total proviral load has not been deter- strated, particularly for asymptomatic carriers (17).
mined. However, multiple clonal expansions of HTLV-1-in- If proviral load were maintained by reverse transcription, fected lymphocytes have been demonstrated for patients with sustained inhibition of reverse transcriptase (RT) may be ex- HAM/TSP and for asymptomatic carriers (2). Consequently, pected to reduce proviral load, by analogy with human immu- Wattel et al. have hypothesized that lymphocyte proliferation nodeficiency virus (HIV) infection (1). In addition to its proven in vitro and in vivo efficacy against HIV, zidovudine has been shown previously to be effective against murine (21) (Rauscher murine leukemia virus) and feline (26) (feline leu- * Corresponding author. Mailing address: Department of Genito- kemia virus) retroviruses in vitro and in vivo.
Urinary Medicine and Communicable Diseases, Imperial College Matsushita et al. (14) found a profound suppression of School of Medicine, Norfolk Place, London W2 1PG, United King- HTLV-1 Gag protein production and a reduction in proviral dom. Phone and fax: 44 171 886 6738. E-mail: [email protected].
† Present address: Institute for Advanced Study, Princeton, NJ DNA when primary CD4⫹ T lymphocytes were exposed to HTLV-1 and cultured in the presence of zidovudine. Nusinoff- TAYLOR ET AL.
TABLE 1. Characteristics of subjectsa antibody titerc assay resultb 100 PBMCsd a Abbreviations: ID, identifier; F, female; M, male; AC, Afro-Caribbean; Cauc, Caucasian; CSF, cerebrospinal fluid; NA, not available.
b The assay used was Genelabs HTLV-I/II 2.3 (Genelabs, Singapore, Singapore).
c The assay used was the Serodia HTLV-1 gel particle agglutination assay (Fujirebio, Tokyo, Japan).
d Median number of HTLV-1 viral DNA copies prior to any antiviral treatment.
Lehrman et al. (18) also found that at zidovudine concentra- Quantification of CTL frequency. CTL effector frequency was determined in a
tions of 2.4 g/ml or above, HTLV-1-specific DNA could not limiting dilution assay (LDA) (4). CD8⫹ lymphocytes, separated from fresh be detected by Southern blot analysis when infected lympho- PBMCs with M-450 anti-CD8-coated Dynabeads, were plated out at known cell cytes were cocultured with susceptible target cells. Macchi et numbers per well in 96-well plates. The CD8⫹ cells were stimulated with phy- tohemagglutinin (1 g/ml) in culture medium (RPMI 1640 medium, 10% fetal al. (13) have shown that low concentrations (as low as 0.1 M) calf serum, and 10% Lymphocult-T [Biotest UK Ltd., Solihull, United King- of zidovudine inhibit transmission of HTLV-1 to adult PBMCs dom]) on day 3. On day 7, the cells were divided into three duplicate 96-well in vitro, inhibiting the production of viral DNA and RNA.
plates and were made up to 200 l/well with the same culture medium. LDAs Zidovudine decreased CD25 expression on T lymphocytes in were performed on day 10 with 3,000 51Cr-labelled, peptide-pulsed autologous culture exposed to HTLV-1 as well as in uninfected PBMCs, Epstein-Barr virus-transformed B cells (target cells) per well. Target cells were pulsed for 1 h with 50 M peptide X1 (MEPTLGQHLPTLSFPD) or X6 but down-regulation of HLA-DR expression was more marked (VIFCHPGQLGAFLTN), which contained the immunodominant Tax epitopes in HTLV-1-infected cells. Zidovudine had no antiretroviral recognized by patient TAN's CTLs (data not shown). Target cells pulsed with effect on PBMCs already infected with HTLV-1 (13). In rab- medium alone were used as controls to measure nonspecific lysis. Lysis in each bits, the administration of zidovudine inhibited HTLV-1 rep- well was considered positive if the 51Cr release exceeded the mean ⫹ 3 standard lication following inoculation of an HTLV-1-transformed cell deviations of the nonspecific lysis. The effector frequencies for both X1 and X6 were added at each time point and represent the minimum number of Tax- line (10). The cytosine analogue 2⬘,3⬘-dideoxycytidine (zalcit- specific CTLs in each sample. The frequency of activated CTLs measured in the abine) also inhibited the synthesis of HTLV-1 viral DNA in LDA correlated closely with the activity of CTLs in the bulk CTL assay (data not CD4⫹ lymphocytes in vitro (14). Two groups who had previ- shown) and was independent of the background lysis.
ously used zidovudine to treat patients with HAM/TSP did not Lymphocyte half-life. To determine the lymphocyte half-life, 10 ml of periph-
report HTLV-1 proviral load (8, 24).
eral blood at each time point was taken into preservative-free sodium heparin (20 IU/ml; Monoparin; CP Pharmaceuticals, Barnstable, United Kingdom) before In order to elucidate the extent of HTLV-1 replication in and after computerized tomography of patient TAN's thorax, which was per- vivo, we studied the effect of nucleoside analogue RT inhibi- formed (for a clinical indication) during the 6th week of therapy with lamivudine tors on the quantity of HTLV-1 viral DNA in PBMCs, T- (week 186), which coincided with the nadir in HTLV-1 DNA copies and anti- lymphocyte phenotype, anti-Tax cytotoxic T-lymphocyte HTLV-1 Tax CTL frequency. The lymphocytes were separated as described (CTL) precursor frequency, and clinical status, first in a patient above and then cultured for 48 h in RPMI 1640 medium with 15% bovine serum (Serum Supreme; BioWhittaker, Walkersville, Md.) and 10 g of lectin (Sigma with early, progressing HAM/TSP and subsequently in four Chemical Co., St. Louis, Mo.) per ml in a conical flask. After disaggregation by other patients with HAM/TSP and high HTLV-1 viral DNA pipetting, the cells were incubated with 0.05 g of N-deacetyl-N-methylcolchicine copy number.
(Demecolcine; Sigma-Aldrich Co. Ltd., Poole, United Kingdom) per ml at 37°C for 30 to 180 min, pelleted (1,000 ⫻ g for 5 min), and resuspended in 10 ml of 0.075 M KCl for 0 to 15 min at room temperature. Cells were again centrifuged (1,000 ⫻ g for 5 min), resuspended gently in 10 ml of freshly prepared 3:1 MATERIALS AND METHODS
methanol-glacial acetic acid, and stored at 4°C. Chromosome preparations were made by dropping the fragile cells onto chilled glass slides and staining them with Subjects. The subjects were five patients with HAM/TSP, whose demographic
3% Giemsa stain for 4 min. Cells (n ⫽ 200) were examined for dicentric chro- and clinical characteristics are shown in Table 1; all cases satisfied the interna- mosomes by a blinded observer (C.A.M.).
tional criteria for diagnosis of HAM/TSP (34). Four patients were ambulant Lymphocyte phenotyping. The phenotype of freshly isolated PBMCs was de-
with aid, and one was wheelchair bound. Four had stable disease for more than termined by flow cytometry (FACScan; Becton Dickinson, Oxford, United King- 3 years, while one, patient TAN, developed HAM/TSP while participating in dom) in the lymphocyte gate with a panel of monoclonal antibodies (MAbs) a prospective study of initially asymptomatic HTLV-1 carriers (28). Following a specific to CD3, CD4, CD8, CD25, CD38, and HLA-DR (Becton Dickinson). A period of assessment with multiple baseline measurements, treatment with a fluorescein isothiocyanate-conjugated anti-mouse immunoglobulin F(ab) nucleoside analogue RT inhibitor was initiated. Patient TAN was treated first ment was used as the second layer. In some assays, directly conjugated phyco- with zidovudine (200 mg) three times daily for 3 months and subsequently with erythrin- and fluorescein isothiocyanate-conjugated MAbs were used. To avoid lamivudine (150 mg) twice daily; the other four patients were treated only with lamivudine (150 mg) twice daily. The mean period of treatment with lamivudine nonspecific binding, the Fc receptor was blocked with human AB serum in all was 10.2 months (range, 5.6 to 17.2 months). No other treatment was taken washes and incubations. Control preparations included omission of the first-layer concurrently, with the single exception of patient TAF, who was undergoing MAb and its replacement by a mouse immunoglobulin of a matched isotype.
treatment with alpha interferon for hepatitis C virus-related chronic active hep- Results were evaluated on a FACScan flow cytometer with a lymphocyte gate.
atitis. The study was approved by the local ethical review board, and all study RT gene sequencing. To search for mutations in the HTLV-1 pol gene that
participants gave written informed consent.
might confer resistance to lamivudine, we determined the sequence of a 2,843-bp Quantification of HTLV-1 viral DNA in PBMCs. To quantify HTLV-1 DNA in
region of HTLV-1 viral DNA encompassing the full coding region of RT and the PBMCs, DNA was extracted from 2 ⫻ 106 PBMCs by the proteinase K method.
initial 909 bp of INT. DNA was extracted from patient TAN's PBMCs before Replicate serial dilutions of the DNA were amplified by a nested-PCR technique treatment, during rebound of viral DNA while the patient was on lamivudine that reliably detected a single copy of HTLV-1 tax DNA in genomic DNA from treatment, and when the viral DNA load had returned to baseline levels after 24 105 cells (29). The viral DNA copy number was calculated from the Poisson weeks of lamivudine therapy. The sequence of PCR-amplified DNA was deter- distribution of negative samples at the cutoff dilution. Interassay variability (0.3 mined, by using an ABI 377 automated DNA sequencer, in forward and reverse log10) was determined by repeated testing of a random selection of patient directions, by direct sequencing without cloning; Taq polymerase errors should samples (29).
therefore make a negligible contribution to the sequence obtained.
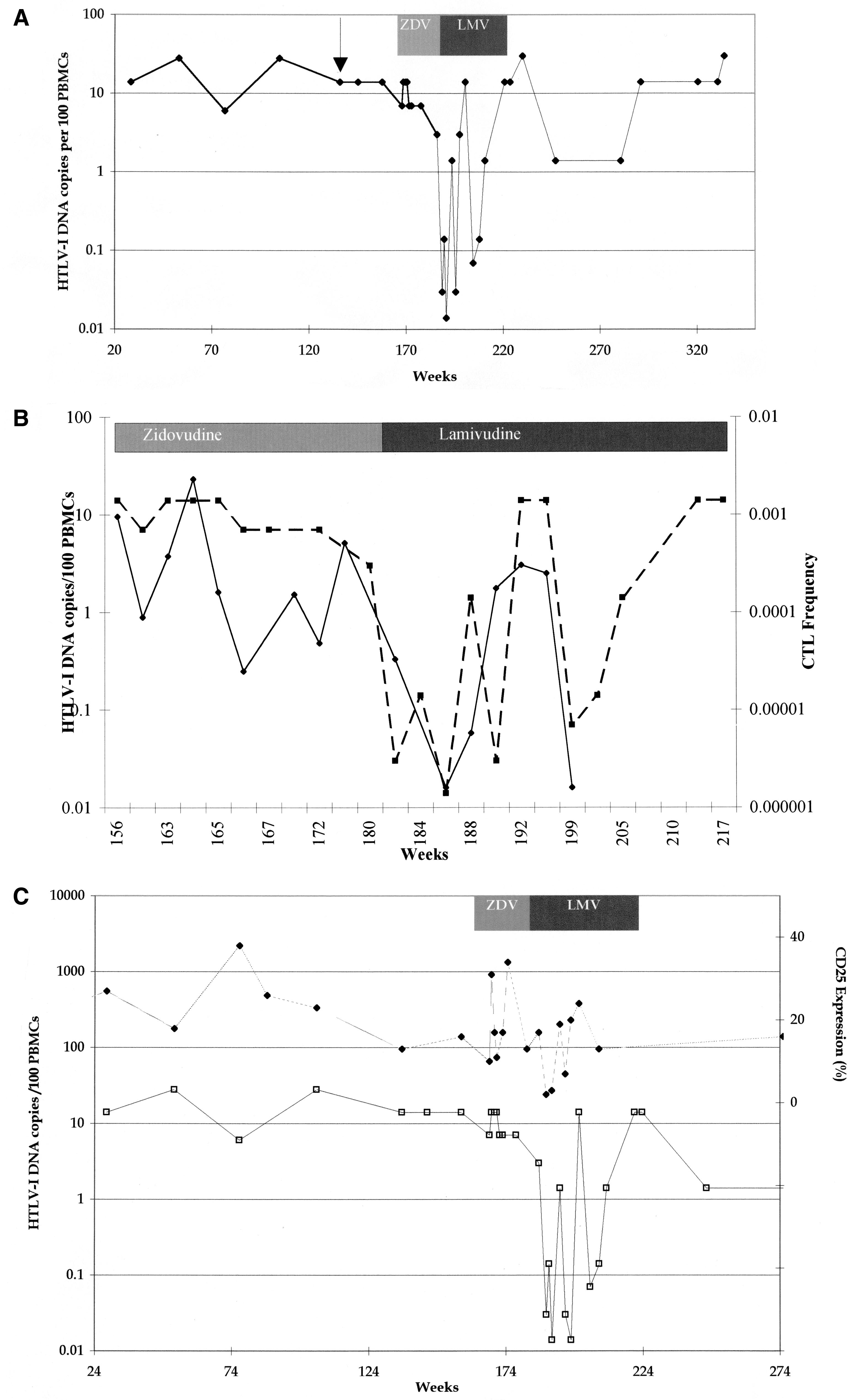
LAMIVUDINE AND HTLV-1 FIG. 1. All patients. (A) Median changes in HTLV-1 DNA copy number for five HTLV-1-infected subjects treated with lamivudine over 24 weeks. (B) Max- imum reduction (nadir) in viral DNA copy number during lamivudine therapy in five patients with HAM/TSP: TAD (solid line with solid squares), TAL (short- dashed line with open squares), TAK (long-dashed line with open squares), TAF (dashed line with open circles), and TAN (shaded line with open triangle).
months' treatment with the RT inhibitor zidovudine (median, 10.5 copies/100 PBMCs; range, 3 to 14 copies) (Fig. 2A). Dur- ing the first 3 weeks of therapy with lamivudine (weeks 180 to 183), HTLV-1 viral DNA copy number fell from 3 to 0.03 per 100 PBMCs (Fig. 2A). While patient TAN was on continuous therapy with lamivudine, viral DNA load rose to baseline levels after 8 weeks and then fluctuated between baseline and nadir over 3 orders of magnitude for the full 24-week duration of therapy. After cessation of lamivudine, the fluctuations be- came smaller and the viral DNA load remained consistently above 1 copy per 100 PBMCs for a further 70 weeks (Fig. 2A).
The rate of HTLV-1 viral DNA decline after the initiation of lamivudine indicated a viral DNA half-life of about 3.2 days.
The estimated total number of HTLV-1-infected PBMCs prior to lamivudine treatment was 3 ⫻ 107. The observed half-life suggests that approximately 6 ⫻ 106 new cells become infected The consensus sequence of RT after 14 weeks of lamivudine therapy (week 201) did not differ from the pre-lamivudine The change in HTLV-1 DNA copies per 100 PBMCs in all (pre-zidovudine and during zidovudine) treatment sequences.
five patients, in relation to duration of therapy with lamivu- However, the consensus RT sequence after 36 weeks of lami- dine, is shown in Fig. 1A. The median pretreatment load was vudine therapy (week 224) differed at 11 nucleotide positions 2.2 HTLV-1 DNA copies/100 PBMCs. The median reduction from the pretreatment sequences (Table 2). Of these 11 se- in viral DNA copy number of all five patients was 1.1 log quences, only one (A3982T) changed the predicted amino acid median nadir in viral DNA load from pretreatment levels was sequence of RT (Q488H); the new amino acid residue (H488) is present in seven of nine HTLV-1 sequences in the GenBank- 10 but the time to reach the nadir varied between patients from 4 to 24 weeks (Fig. 1B).
EMBL database and is therefore unlikely to have been se- Patient TAN had been monitored prospectively over a pe- lected in patient TAN because it conferred resistance to lami- riod of 3 years prior to and during the development of HAM/ vudine. In particular, no YMDD substitution was found. One TSP; the HTLV-1 DNA copy number in PBMCs was consis- further silent nucleotide substitution was found in the inte- tently high over this period, with a median of 14 (range, 7 to grase coding region (T4588C); again, this substitution ap- 28) copies of viral DNA per 100 PBMCs, compared with other peared for the first time after 24 weeks of lamivudine therapy.
asymptomatic carriers (median viral DNA, 0.28 copies per 100 Prior to treatment, a high frequency of activated anti-Tax PBMCs) (28), and this was not significantly reduced during 3 CD8⫹ lymphocytes recognizing several Tax epitopes simulta-
TAYLOR ET AL.
LAMIVUDINE AND HTLV-1
neously was present in the peripheral circulation of patient
TABLE 2. Effect of treatment on HTLV-1 DNA sequencea
TAN. During treatment with lamivudine, the anti-Tax CTL
frequency declined with the fall in viral DNA load and then
rose again as the viral DNA copy number increased (Fig. 2B).
The frequency of anti-Tax CTLs prior to lamivudine treatment
was 7.9 ⫻ 10⫺4 CD8⫹ cells, declining to 1.6 ⫻ 10⫺6 CD8⫹ cells
after 5 weeks of therapy. This is consistent with a CTL half-life
of 3.9 days. Since the LDA is conducted over a period equal to
2.5 in vivo half-lives of the CTLs, the results may underesti-
mate the frequency of Tax-specific CTLs; however, in vitro
their half-life may be prolonged by the exogenous interleukin-2
Prospective quantification of T-lymphocyte phenotypic
markers was undertaken with patient TAN. There was no
trend in the absolute counts or percentage of CD4⫹ or CD8⫹
T-lymphocyte counts or in the absolute number of circulating
a Sequencing revealed the emergence of a new dominant HTLV-1 DNA se-
lymphocytes over time and no change in relation to lamivudine
quence with 11 nucleotide substitutions in the consensus sequence of the
therapy. However, the proportion of CD25⫹ lymphocytes,
HTLV-1 pol gene from patient TAN. Each of these substitutions was first
which was elevated before and after lamivudine therapy, fell
identified at the final time point, after 36 weeks of treatment with lamivudine,
from 15 to 2%, coincident with the reduction in viral DNA
with the reexpansion of the amount of viral DNA. Before treatment, the nucle-
otide present at each of these positions was the same as that in the reference
copies on the initiation of lamivudine therapy and fluctuated in
sequence (stub) (23). —, no amino acid change.
a manner parallel to that of the viral DNA copy number and
anti-Tax CTL precursor frequency changes during therapy
(Fig. 2C). The estimated half-life of CD25⫹ lymphocytes was
The 50% infective dose of lamivudine in PBMCs was 2,533 M
The presence of both abundant anti-Tax CTLs and the high
(compared to 90 M for zidovudine). Similar results were
viral DNA load suggested the possibility that there might be
obtained for lymphocyte cell lines including HTLV-1 provirus-
frequent CTL-mediated killing of infected CD4⫹ T lympho-
bearing C8166 cells. The administration of lamivudine orally,
cytes in vivo. Lymphocyte half-lives can be estimated by mea-
in the dose used in this study, results in a maximum concen-
suring the rate of disappearance from the circulation of lym-
tration of lamivudine in serum which is 6 log10 below the
phocytes with stable, radiation-induced chromosome damage
(15). We therefore counted the proportion of PBMCs that
Initiation of lamivudine therapy was followed by an imme-
contained dicentric chromosomes, after patient TAN had un-
diate reduction in viral DNA copy number. Subsequently, the
dergone computerized tomography of the thorax, and their
viral DNA copy number oscillated, with the troughs gradually
rate of disappearance from the blood (Fig. 3). The estimated
rising toward the baseline. CTL precursor frequency and
half-life of 2.5 days reflects the rate at which the PBMCs were
CD25⫹ T lymphocytes also showed periodic oscillations during
dividing or dying and is consistent with the observed rapid
lamivudine therapy, which appeared to coincide with the
reduction in HTLV-1 viral DNA (estimated half-life, 3.2 days)
changes in viral DNA load. Mathematical analysis shows that
and CTL frequency (estimated half-life, 3.9 days).
such oscillations in the quantity of viral DNA may result from
The only clinical improvement was seen in the patient with
the production of HTLV-1-infected cells from both cell divi-
recent-onset HAM/TSP, an improvement which persisted only
sion (not affected by lamivudine) and reverse transcription and
during the period when lamivudine appeared to have reduced
from the interplay of viral production with CTL-mediated kill-
the viral burden. The patients with severe, long-standing, and
ing of infected cells (33). This model suggests that the high
stable disability showed no symptomatic improvement despite
viral DNA load of HTLV-1 is indeed maintained by proviral
reduced HTLV-1 DNA load (27).
expansion but that this is consistent with, and indeed may
require, a high rate of HTLV-1 virion production (33). The
subsequent return to pretreatment viral DNA levels in a com-
pliant patient could be due to cellular resistance (a reduction
In this study, zidovudine treatment of patient TAN led to no
in the uptake or phosphorylation of lamivudine) or the emer-
change in HTLV-1 viral DNA copy number, CTL precursor
gence of viral variants with decreased susceptibility to lamivu-
frequency, or T-lymphocyte phenotype. Treatment was there-
dine. Lamivudine resistance in HIV-1 infection is often accom-
fore changed to lamivudine, a cytosine analogue which has
panied by a mutation in the active site of the HIV-1 RT (22).
been found to be safe and effective with patients infected with
However, the corresponding region in the HTLV-1 RT gene
HIV-1 (31) or hepatitis B virus (6). Other licensed nucleoside
was unchanged for patient TAN. The only coding change that
analogues, didanosine, stavudine, and zalcitabine, are associ-
appears in the RT gene following lamivudine treatment is
ated with peripheral neuropathy during prolonged therapy
commonly found in naturally occurring isolates of HTLV-1
(25). In cell proliferation assays (Glaxo Research and Devel-
and is therefore unlikely to have been selected because it
opment Ltd.) (12a), lamivudine does not affect the uptake of
confers resistance to lamivudine. However, the de novo ap-
[3H]thymidine by uninfected and HTLV-1-infected PBMCs.
pearance of 11 nucleotide substitutions in the consensus se-
FIG. 2. Patient TAN. (A) HTLV-1 viral DNA copy numbers from all available time points and their relationship to the onset of HAM/TSP (arrow) and therapy
first with zidovudine (ZDV) and then with lamivudine (LMV) are shown for patient TAN. (B) Comparison of changes in the amount of HTLV-1 viral DNA (dashed
line with squares) and HTLV-1 Tax-specific CTL effector frequency (solid line with diamonds) during therapy. (C) Comparison of the amount of HTLV-1 viral DNA
(open squares) with changes in the percentage of lymphocytes expressing CD25 (solid diamonds) before, during, and after therapy with lamivudine. ZDV, zidovudine;
LMV, lamivudine.
TAYLOR ET AL.
average life expectancy of patient TAN's PBMCs of 3 to 6 days.
CTL-mediated killing of HTLV-1-infected cells may account
for part of this rapid cell turnover. However, other factors must
also have been involved, as even rapid killing of a minority
population of PBMCs expressing Tax protein (at most, 14% of
PBMCs) and high turnover of CD8⫹ HTLV-specific CTLs
(which may constitute up to 10% of CD8⫹ PBMCs [16a])
would not reduce the average life expectancy of all PBMCs so
far below the normal value of approximately 150 days (memory
lymphocytes) or 3.5 years (naive lymphocytes) (15). Vigorous
cell activation and proliferation, driven by HTLV-1 Tax pro-
tein, may have predisposed PBMCs to apoptosis (3).
In conclusion, lamivudine reduced the amount of HTLV-1
DNA in five of five patients with HAM/TSP. This reduction
and the subsequent increase in the amount of viral DNA dur-
ing treatment are consistent with active viral replication
through reverse transcription. The viral kinetics indicate that
the importance of viral replication in maintaining viral DNA
load varies between patients. The results have important im-
plications for our understanding of HTLV-1 pathogenesis and
might open the way to specific therapy for HTLV-1-associated
FIG. 3. No dicentric chromosomes were found in cell preparations before
computerized tomography (CT), 4.5% of cells contained dicentric chromosomes
48 h after computerized tomography, 2% dicentric chromosomes were detected
S.E.H. and C.R.M.B. are supported by The Wellcome Trust. G.P.T.
96 h post-computerized tomography, and none were detected at 336 h. No
was supported by the Jefferiss Trust. The HTLV European Research
reduction in the total lymphocyte count occurred during this period. Solid dia-
Network supported by the European Community Biomed Programme
monds, observed rate of disappearance of dicentric chromosomes for patient
TAN. Open squares, rate of disappearance of dicentric chromosomes following
(BMH4 CT98-3781) provided a forum for critical appraisal of this
radiotherapy for ankylosing spondylitis (15). The lymphocyte half-life is about
Jennifer Tosswill of the Central Public Health Laboratory, Colin-
dale, London, United Kingdom, assisted with the HTLV-1 DNA mea-
quence of a 2,843-bp region of the HTLV-1 genome strongly
suggests that the virus is replicating, causing the introduction
1. Brun-Vezinet, F., C. Boucher, C. Loveday, D. Descamps, V. Fauveau, J.
of polymerase errors, and that the viral population passed
Izopet, D. Jeffries, S. Kaye, C. Krzyanowski, A. Nunn, R. Schuurman, J. M.
through a bottleneck. The newly emergent consensus sequence
Seigneurin, C. Tamalet, R. Tedder, J. Weber, and G. J. Weverling. 1997.
HIV-1 viral load, phenotype, and resistance in a subset of drug-naive par-
represents a clone that (by chance) survived the genetic bot-
ticipants from the Delta trial. The National Virology Groups. Delta Virology
tleneck and grew to replace the preexisting dominant se-
Working Group and Coordinating Committee. Lancet 350:983–990.
quence. Although we cannot exclude the possibility of drug
2. Cavrois, M., A. Gessain, S. Wain-Hobson, and E. Wattel. 1996. Proliferation
resistance mutations in a minority of viruses, any such viruses
of HTLV-1 infected circulating cells in vivo in all asymptomatic carriers and
patients with TSP/HAM. Oncogene 12:2419–2423.
cannot have made a major contribution to the recrudescence
3. Chlichlia, K., G. Moldenhauer, P. T. Daniel, M. Busslinger, L. Gazzolo, V.
of HTLV-1 viral DNA in PBMCs.
Schirrmacher, and K. Khazaie. 1995. Immediate effects of reversible
Our previous finding of abundant activated anti-Tax CTLs in
HTLV-I tax function: T-cell activation and apoptosis. Oncogene 10:269–277.
HTLV-1-infected patients (4) already suggested that there is
4. Daenke, S., A. Kermonde, S. E. Hall, G. Taylor, J. Weber, S. Nightingale,
and C. R. M. Bangham. 1996. High activated and memory cytotoxic T-cell
persistent expression of the tax gene in the infected host. How-
responses to HTLV-1 in healthy carriers and patients with tropical spastic
ever, it was not clear whether expression was confined to the
paraparesis. Virology 217:139–146.
tax gene or whether it represented persistent replication of
5. Dalgleish, A., J. Richardson, E. Matutes, K. Cruickshank, A. Newell, A.
HTLV-1. The rapid fall in viral DNA for three patients during
Sinclair, R. Thorpe, M. Brasher, J. Weber, D. Catovsky, et al. 1989. HTLV-I
infection in tropical spastic paraparesis: lymphocyte culture and serological
treatment with lamivudine suggests that the high pretreatment
response. AIDS Res. Hum. Retroviruses 4:475–485.
viral DNA copy number was maintained chiefly by active viral
6. Dienstag, J. L., R. P. Perrillo, E. R. Schiff, M. Bartholomew, C. Vicary, and
replication. Proliferation of infected lymphocytes, which has
M. Rubin. 1995. Preliminary trial of lamivudine for chronic hepatitis B
been suggested by others to be the main cause of high proviral
infection. N. Engl. J. Med. 333:1657–1661.
7. Gessain, A., J. C. Vernant, L. Maurs, F. Barin, O. Gout, A. Calender, and G.
load in HTLV-1 infection, is not sufficient to maintain the high
de The´. 1985. Antibodies to human T-lymphotropic virus type-I in patients
viral DNA load in these patients. These results are in keeping
with tropical spastic paraparesis. Lancet ii:407–409.
with the recent observation that HTLV-1 mRNA was more
8. Gout, O., A. Gessain, M. Iba-Zizen, S. Kouzan, F. Bolgert, G. de The´, and O.
frequently detected in patients with a high proviral load (19).
Lyon-Caen. 1991. The effect of zidovudine on chronic myelopathy associated
with HTLV-I. J. Neurol. 238:108–109.
However, for two patients a much more gradual decline in the
9. International Agency for Research on Cancer. 1996. IARC monographs on
amount of viral DNA was noted, suggesting that the relative
the evaluation of carcinogenic risks to humans, vol. 67. Human immunode-
importance of reverse transcription in the maintenance of viral
ficiency viruses and human T-cell lymphotrophic viruses. International
DNA load differs between subjects.
Agency for Research on Cancer, Lyon, France.
10. Isono, T., K. Ogawa, and A. Seto. 1990. Antiviral effect of zidovudine in the
Four independent techniques (limiting dilution analysis of
experimental model of adult T-cell leukaemia in rabbits. Leuk. Res. 14:841–
CTL frequency, HTLV-1 viral DNA quantification, fluores-
cence-activated cell sorting analysis of lymphocyte phenotypes,
11. Jacobson, S., H. Shida, D. E. McFarlin, A. S. Fauci, and S. Koenig. 1990.
and the clearance of cells containing dicentric chromosomes)
Circulating CD8⫹ cytotoxic T lymphocytes specific for HTLV-I pX in pa-
tients with HTLV-I associated neurological disease. Nature 348:245–248.
during antiretroviral therapy each produced estimates of the
12. Koralnik, I. J., E. Boeri, W. C. Saxinger, A. Lo Monico, J. Fullen, A. Gessain,
LAMIVUDINE AND HTLV-1
H.-G. Guo, R. C. Gallo, P. Markham, V. Kalyanaraman, V. Hirsch, J. Allen,
Rapid changes in human immunodeficiency virus type 1 RNA load and
K. Murthy, P. Alford, J. P. Slattery, S. J. O'Brien, and G. Franchini. 1994.
appearance of drug resistant virus populations in persons treated with lami-
Phylogenetic associations of human and simian T-cell leukemia/lympho-
vudine (3TC). J. Infect. Dis. 171:1411–1419.
tropic virus type I strains: evidence for interspecies transmission. J. Virol.
23. Seiki, M., S. Hattori, Y. Hirayama, and M. Yoshida. 1983. Human adult
T-cell leukemia virus: complete nucleotide sequence of the provirus genome
12a.Macchi, B. Personal communication.
integrated in leukemia cell DNA. Proc. Natl. Acad. Sci. USA 80:3618–3622.
13. Macchi, B., I. Faraoni, J. Zhang, S. Grelli, C. Favalli, A. Mastino, and E.
24. Sheremata, W. A., D. Benedict, D. C. Squilacote, A. Sazant, and S. DeFrei-
Bonmassar. 1997. AZT inhibits the transmission of human T cell leukaemia/
tas. 1993. High-dose zidovudine induction in HTLV-I-associated myelopa-
lymphoma virus type I to adult peripheral blood mononuclear cells in vitro.
thy: safety and possible efficacy. Neurology 43:2125–2129.
J. Gen. Virol. 78:1007–1016.
25. Simpson, D. M., and M. Tagliati. 1995. Nucleoside analogue-associated
14. Matsushita, S., H. Mitsuya, M. S. Reitz, and S. Broder. 1987. Pharmacolog-
peripheral neuropathy in human immunodeficiency virus infection. J. Ac-
ical inhibition of in vitro infectivity of human T lymphotropic virus type I. J.
quir. Immune Defic. Syndr. Hum. Retrovirol. 9:153–161.
Clin. Investig. 80:394–400.
26. Tavares, L., C. Roneker, K. Johnston, S. N. Lehrman, and F. de Noronha.
15. McLean, A. R., and C. A. Michie. 1995. In vivo estimates of division and
1987. 3⬘-Azido-3⬘-deoxythymidine in feline leukemia virus-infected cats: a
death rates of human T lymphocytes. Proc. Natl. Acad. Sci. USA 92:3707–
model for therapy and prophylaxis of AIDS. Cancer Res. 47:3190–3194.
27. Taylor, G. P., S. Hall, M. Nowak, C. Michie, M. Rossor, R. Davis, C. R. M.
16. Nagai, M., K. Usuku, W. Matsumoto, D. Kodama, N. Takenouchi, T. Mori-
Bangham, and J. N. Weber. 1998. Reduction of HTLV-I proviral load in
toyo, S. Hashiguchi, M. Ichinose, C. R. M. Bangham, S. Izumo, and M.
patients with HTLV-I associated myelopathy through lamivudine mono-
Osame. 1998. Analysis of HTLV-I proviral load in 202 HAM/TSP patients
therapy, abstr. 508, p. 175. In Abstracts of the 5th Conference on Retrovi-
and 243 asymptomatic HTLV-I carriers: high proviral load strongly predis-
poses to HAM/TSP. J. Neurovirol.
ruses and Opportunistic Infections, Chicago, Ill., 1 to 5 February 1998.
Navarrete, S., S. E. Hall, and C. R. M. Bangham. Unpublished data.
Taylor, G. P., J. H. C. Tosswill, E. Matutes, S. Daenke, S. Hall, B. Bain, M.
17. Niewiesk, S., S. Daenke, C. E. Parker, G. P. Taylor, J. N. Weber, S. Night-
Rossor, D. Thomas, C. R. M. Bangham, and J. N. Weber. 1999. Inflammatory
ingale, and C. R. M. Bangham. 1994. The transactivator gene of human
consequences of HTLV-I infection in an initially asymptomatic UK cohort.
T-cell leukemia virus type I is more variable within and between healthy
J. Acquir. Immune Defic. Syndr. Hum. Retrovirol. 22:92–100.
carriers than patients with tropical spastic paraparesis. J. Virol. 68:6778–
29. Tosswill, J. H. C., G. P. Taylor, J. P. Clewley, and J. N. Weber. 1998.
Quantification of proviral DNA load in human T-cell leukaemia virus type-I
18. Nusinoff-Lehrman, S., M. St. Clair, R. L. Miller, S. Broder, H. R. Wilson, M.
infections. J. Virol. Methods 75:21–26.
Bushby, et al. 1986. Azidothymidine: spectrum of in vitro antimicrobial
30. Uchiyama, T., J. Yodoi, K. Sagawa, K. Takatsuki, and H. Uchino. 1977.
activity, abstr. 556, p. 66. In Proceedings of the Second International Con-
Adult T-cell leukaemia: clinical and haematological features of 16 cases.
ference on AIDS, Paris, France, 23 to 25 June 1986.
19. Okayama, A., N. Tachibana, S. Ishihara, Y. Nagatomo, K. Murai, M. Oka-
31. van-Leeuwen, R., C. Katlam, V. Kitchen, C. A. Boucher, R. Tubiana, M.
moto, T. Shima, K. Sagawa, H. Tsubouchi, S. Stuver, and N. Mueller. 1997.
McBride, D. Ingrand, J. Weber, A. Hill, H. McDade, et al. 1995. Evaluation
Increased expression of interleukin-2 receptor alpha on peripheral blood
of safety and efficacy of 3TC (lamivudine) in patients with asymptomatic or
mononuclear cells in HTLV-I tax/rex mRNA-positive asymptomatic carriers.
mildly symptomatic human immunodeficiency virus infection: a phase I/II
J. Acquir. Immune Defic. Syndr. Hum. Retrovirol. 15:70–75.
study. J. Infect. Dis. 171:1166–1171.
20. Osame, M., K. Usuku, S. Izumo, N. Ijichi, H. Amitani, A. Igata, M. Matsu-
32. Wattel, E., M. Cavrois, A. Gessain, and S. Wain-Hobson. 1996. Clonal
moto, and M. Tara. 1986. HTLV-I associated myelopathy, a new clinical
expansion of infected cells. A way of life for HTLV-I. J. Acquir. Immune
entity. Lancet i:1031–1032. (Letter.)
Defic. Syndr. Hum. Retrovirol. 13(Suppl. 1):S92–S99.
21. Ruprecht, R. M., L. G. O'Brien, L. D. Rossoni, and S. Nusinoff-Lehrman.
33. Wodarz, D., M. A. Nowak, and C. R. M. Bangham. 1999. The dynamics of
1986. Suppression of mouse viraemia and retroviral disease by 3⬘-azido-3⬘-
HTLV-I and the CTL response. Immunol. Today 20:220–227.
deoxythymidine. Nature 323:467–469.
34. World Health Organization. 1989. WHO diagnostic guidelines of HAM.
22. Schuurman, R., M. Nijhuis, L. M. van-Leeuwen, P. Schipper, D. de-Jong, P.
Virus diseases. Human T-lymphotropic virus type I, HTLV-I. Weekly Epi-
Collis, S. A. Danner, J. Mulder, C. Loveday, C. Christopherson, et al. 1995.
demiol. Rec. 49:382–383.
Source: http://ped.fas.harvard.edu/files/ped/files/jvirol99b_0.pdf
The dubai logistics cluster
THE DUBAI LOGISTICS CLUSTER Alanood Bin Kalli, Camila Fernandez Nova, Hanieh Mohammadi, Yasmin Sanie-Hay, Yaarub Al Yaarubi COUNTRY OVERVIEW The United Arab Emirates (UAE) is a federation of seven emirates, each governed by its own monarch. The seven Emirates - Abu Dhabi, Ajman, Dubai, Fujairah, and- jointly form the Federal Supreme Council, which chooses a president
boletincipei.unanleon.edu.ni
Boletín Electrónico sobre Integración Regional del CIPEI ISSN: 2223-2117, Vol. 3, mayo de 2013 «Actos Unilaterales Internacionales: ¿Nicaragua tiene Derecho al pago de la indemnización contra Estados Unidos derivada de la sentencia de la CIJ del 27 de Junio de 1986? » Marisol Victoria Ruiz Mendoza y Andrés Noé Urbina Munguía