Encapsulation of amoxicillin within laponite-doped poly(lactic-<italic>co</italic>-glycolic acid) nanofibers: preparation, characterization, and antibacterial activity

Encapsulation of Amoxicillin within Laponite-Doped Poly(lactic-co-glycolic acid) Nanofibers: Preparation, Characterization, and
Antibacterial ActivityShige Wang,Fuyin Zheng,Yunpeng Huang,Yuting Fang,Mingwu Shen,Meifang Zhu,and Xiangyang Shi†State Key Laboratory for Modification of Chemical Fibers and Polymer Materials, Donghua University, Shanghai 201620, People's
Republic of China
‡College of Materials Science and Engineering, Donghua University, Shanghai 201620, People's Republic of China
§College of Chemistry, Chemical Engineering and Biotechnology, Donghua University, Shanghai 201620, People's Republic of China
⊥CQM-Centro de Química da Madeira, Universidade da Madeira, Campus da Penteada, 9000-390 Funchal, Portugal
ABSTRACT: We report a facile approach to encapsulatingamoxicillin (AMX) within laponite (LAP)-doped poly(lactic-co-glycolic acid) (PLGA) nanofibers for biomedical applica-tions. In this study, a synthetic clay material, LAP nanodisks,was first used to encapsulate AMX. Then, the AMX-loadedLAP nanodisks with an optimized AMX loading efficiency of9.76 ± 0.57% were incorporated within PLGA nanofibersthrough electrospinning to form hybrid PLGA/LAP/AMXnanofibers. The loading of AMX within LAP nanodisks and theloading of LAP/AMX within PLGA nanofibers werecharacterized via different techniques. In vitro drug release profile, antimicrobial activity, and cytocompatibility of the formedhybrid PLGA/LAP/AMX nanofibers were also investigated. We show that the loading of AMX within LAP nanodisks does notlead to the change of LAP morphology and crystalline structure and the incorporation of LAP/AMX nanodisks does notsignificantly change the morphology of the PLGA nanofibers. Importantly, the loading of AMX within LAP-doped PLGAnanofibers enables a sustained release of AMX, much slower than that within a single carrier of LAP nanodisks or PLGAnanofibers. Further antimicrobial activity and cytocompatibility assays demonstrate that the antimicrobial activity of AMX towardthe growth inhibition of a model bacterium of Staphylococcus aureus is not compromised after being loaded into the hybridnanofibers, and the PLGA/LAP/AMX nanofibers display good cytocompatibility, similar to pure PLGA nanofibers. With thesustained release profile and the reserved drug activity, the organic/inorganic hybrid nanofiber-based drug delivery system may
find various applications in tissue engineering and pharmaceutical science.
KEYWORDS: poly(lactic-co-glycolic acid), laponite, electrospinning, amoxicillin, sustained release, antimicrobial activity
method, drug molecules are directly integrated within the
The distinctive features of nanofibers such as flexibility in
nanofibers by simply electrospinning the drug/polymer mixture
surface functionalities, superior mechanical durability, and
solution or by absorbing/assembling the drugs onto the
interconnected and readily controlled secondary structures
nanofiber surfaces or in the interior of nanofibers−
afford them to be used as a unique drug delivery system, which
Although this method allows easy incorporation of drug
has inherent advantages including easy implementation, little
molecules within the nanofibers, a burst release often occurs,
influence on the drug activity, and well controlled drug release
which is not desirable in most cases.Emulsion and coaxial
rate.−Electrospinning is a simple and straightforward way to
electrospinning are two improved techniques to be used for
produce nanofibers with designed structure and morphol-
drug delivery applications, which is able to mitigate the burst
ogy.Since Kenawy et al. first examined the drug release
release of the drug to some extent.In both methods, the
property from electrospun nanofibers, the use of electrospun
drugs are able to be embedded into the core region of the
nanofibers for drug delivery applications has received increasing
nanofibers in a reservoir-type model and form a so-called
interest in the pharmaceutical fiUntil now, a number ofdifferent drug-loading methods have been developed via
September 27, 2012
conventional, emulsion, or coaxial electrospinning techni-
Accepted: November 6, 2012
ques.−In the conventional single fluid electrospinning
Published: November 6, 2012
2012 American Chemical Society
dx.doi.org/10.1021/am302130b ACS Appl. Mater. Interfaces 2012, 4, 6393−6401

ACS Applied Materials & Interfaces
Scheme 1. Schematic Illustration of the Loading of Free AMX Drug within PLGA Nanofibers (a) and the Loading of AMX/LAPwithin PLGA Nanofibers (b)
"core−sheath" structure. In this reservoir-type structure, the
Staphylococcus aureus (S. aureus) as a model bacterium both in
outer polymer shell can act as an additional barrier to control
liquid and on solid medium. Finally, the cytocompatibility of
the drug release profiNevertheless, there are still some
PLGA/LAP/AMX nanofibers was evaluated through 3-(4,5-
issues existing in the emulsion and coaxial electrospinning
techniques. For example, the coaxial electrospinning may need
colorimetric assay and scanning electron microscopy (SEM)
substantial optimization of the electrospinning parameters, and
morphology observation of porcine iliac artery endothelial cells
the emulsifier used in emulsion electrospinning may cause
(PIEC) cultured onto the nanofiber scaffold. To our knowl-
compromised biocompatibility of the nanofibers.Therefore,
edge, this is the first report related to the development of
development of other nanofiber systems that can overcome the
PLGA/LAP composite nanofibers for drug delivery applica-
burst release of the encapsulated drugs still remains a great
In our previous study, we reported the use of halloysite
■ EXPERIMENTALSECTION
nanotubes (HNTs)/poly(lactic-co-glycolic acid) (PLGA) com-
Materials. PLGA (Mw = 81 000 g/mol) with a lactic acid/glycolic
posite nanofibers for encapsulation and release of a model drug
acid ratio of 50:50 and LAP were purchased from Jinan Daigang
tetracycline hydrochloride In this approach, the TCH
Biotechnology Co., Ltd. (China) and Zhejiang Institute of Geologic
drug molecules were first physically encapsulated within the
and Mineral Resources (China), respectively. AMX was from Shanghai
HNTs, followed by electrospinning the mixture solution of
Yuanye Biotechnology Co., Ltd. S. aureus was purchased from
PLGA and TCH-loaded HNTs to form a composite drug-
Shanghai Fuzhong Biotechnology Development Co., Ltd. Luria-
incorporated nanofiber, which was proven to be able to
Bertani (LB)-medium and agar were from Beijing AoboxingBiotechnology Co., Ltd., tetrahydrofuran (THF), and N,N-dimethyl-
significantly alleviate the burst release of the TCH. This
formamide (DMF) were from Sinopharm Chemical Reagent Co., Ltd.
preliminary success leads us to hypothesize that other naturally
(China). PIEC cells were obtained from Institute of Biochemistry and
occurring or synthetic clay materials that have been used for
Cell Biology (the Chinese Academy of Sciences, Shanghai).
drug delivery applications may also be able to be incorporated
Dulbecco's Modified Eagle's medium (DMEM), fetal bovine serum
within polymer nanofibers to improve the drug release profile
(FBS), penicillin, and streptomycin were purchased from Hangzhou
for various biomedical applications. As a synthetic clay material,
Jinuo Biomedical Technology (Hangzhou, China). All chemicals and
laponite (LAP) has been used as a drug carrier because the
reagents were used as received. Water used in all experiments was
interlayer space of LAP can be used for effective drug
purified using a Milli-Q Plus 185 water purification system (Millipore,
encapsulation with high retention capacity.−For example,
Bedford, MA) with resistivity higher than 18 MΩ·cm.
Preparation of Drug-Loaded LAP Nanodisks. AMX was first
Jung et al. reported the incorporation of a hydrophobic drug
dissolved into water to obtain AMX aqueous solutions with different
itraconazole (ITA) into LAP through an interfacial interaction
concentrations (0.1, 0.3, 0.5, 1, and 2 mg/mL, respectively) at room
of LAP and ITA. However, their release data showed that the
temperature. Then, LAP nanodisks were dispersed into the resulting
release of ITA from ITA/laponite hybrid could reach 75%
AMX solutions with different concentrations (3, 5, and 10 mg/mL,
during the first 24 h.Take the excellent biodegradability,
respectively) to get a batch of LAP/AMX suspensions. After that, the
biocompatibility, and electrospinnability of PLGA into
LAP/AMX suspensions were magnetically stirred for 24 h in order to
account,it is expected that PLGA/LAP composite
make the LAP fully swell and to make the AMX molecules be
nanofibers may be used as drug carriers to afford the
sufficiently intercalated into the interlayer of LAP. The LAP/AMXnanodisks were then separated by centrifugation (8000 rpm, 5 min)
encapsulated drugs with a sustained release profile.
and washed with water for 3 times to remove the excessive AMX. The
In this present study, LAP nanodisks were first used to
supernatants after 4 times centrifugation were collected together, and
encapsulate a model drug of amoxicillin (AMX). Then, the
the nonencapsulated AMX was quantified using a Lambda 25 UV−vis
LAP/AMX nanodisks were incorporated within PLGA nano-
spectrophotometer (Perkin-Elmer, USA) at 230 nm with a
fibers via electrospinning to form PLGA/LAP/AMX nanofibers
concentration−absorbance calibration curve at the same wavelength.
(Scheme ). The formed LAP/AMX nanodisks and the
Finally, the LAP/AMX nanodisks were obtained by lyophilization. The
composite PLGA/LAP/AMX nanofibers were intensively
drug loading efficiency can be calculated from the following equation:
characterized using different techniques. In vitro drug release
loading efficiency = M /M × 100%
behavior of the composite PLGA/LAP/AMX nanofibers was
examined using UV−vis spectroscopy. The antimicrobial
where Mt and M0 stand for the mass of encapsulated AMX and the
activity of the composite nanofibers was investigated using
initial total AMX used for encapsulation, respectively.
dx.doi.org/10.1021/am302130b ACS Appl. Mater. Interfaces 2012, 4, 6393−6401
ACS Applied Materials & Interfaces
Preparation of AMX-Loaded Electrospun Nanofibers. PLGA
fresh PBS solution was replenished. The optical density (OD) value
was dissolved in a mixed solvent of THF/DMF (v/v = 3:1) with an
was measured using a Lambda 25 UV−vis spectrophotometer at 230
optimized concentration of 25% (w/v).After that, AMX (0.5 wt %
relative to PLGA) or LAP/AMX (with final 0.5% AMX relative to
In Vitro Antibacterial Activity Assay. The antibacterial activity
PLGA) was dispersed into PLGA solution for subsequent electro-
of LAP/AMX nanodisks, PLGA/AMX nanofibers, and PLGA/LAP/
spinning to form PLGA/AMX or PLGA/LAP/AMX nanofibers,
AMX nanofibers was evaluated in liquid medium (2.5 g LB medium
respectively (Scheme PLGA/LAP nanofibers without AMX but
dissolved into 100 mL water) by recording the absorbance of the
with the same amount of LAP used to encapsulate 0.5% AMX (relative
solution at 625 nm using a Lambda 25 UV−vis spectrophotometer,
to PLGA) were also prepared as a control material. The electro-
which is in direct proportion to the bacterial number.In brief, 5
spinning system was made up of a syringe pump with a 10 mL syringe,
mL of the bacterial solution with an OD value of 0.1−0.2 at 625 nm
a silicone hose, a stainless steel needle with an inner diameter of 0.8
was added into each 15 mL glass tube. Then, AMX powder, LAP/
mm, a high voltage power supply, and a thin aluminum foil acting as a
AMX nanodisks, PLGA/AMX nanofibers, and PLGA/LAP/AMX
collector which was positioned horizontally and grounded. A clamp
nanofibers were added into each tube with the AMX concentration of
was used to connect the high voltage power supply with the needle.
10, 20, and 30 μg/mL, respectively. AMX powder was used as a
The electrospinning process was carried out under ambient condition
positive control, while PLGA and PLGA/LAP nanofibers without
with a fixed electrical potential of 20 kV, a collect distance of 15 cm,
AMX were used as negative controls. Tube without sample was set as
and a feeding rate of 0.8 mL/h by a syringe pumpAfter
another negative control. All the samples were in triplicate and
electrospinning, nanofibers were taken off from the collector and
incubated at 37 °C with a shaking speed of 100 rpm for 24 h. After
vacuum-dried for at least 48 h to remove the residual organic solvent
that, the OD value at 625 nm was monitored using UV−vis
and moisture.
spectroscopy. The bacterial inhibition percentage can be calculated
Characterization Techniques. The LAP and LAP/AMX nano-
by the following eqution:
disks were characterized using Fourier transform infrared (FTIR)spectroscopy. The analysis was performed using a Nicolet Nexus 670
bacterial inhibition (%) = I
( − I )/I × 100
FTIR (Nicolet-Thermo) spectrometer. All spectra were recorded using
where Ic and Is are the average ODs of the control group and the
a transmission mode with a wavenumber range of 650−4000 cm−1.
experimental group, respectively. The above method was also used to
The morphology of LAP and LAP/AMX nanodisks was observed
evaluate the correlation between the antibacterial activity of the
using field emission scanning electron microscopy (FESEM)
PLGA/LAP/AMX nanofibers as a function of the release time. Briefly,
(HITACHI S-4800, Japan) with an accelerating voltage of 15 kV.
PLGA/LAP/AMX nanofibers with the AMX mass of 100 μg were
The LAP or LAP/AMX nanodisks were first dispersed into water.
added to a glass tube containing 5 mL of the bacterial suspension with
Then, the suspension of LAP or LAP/AMX nanodisks was dropped
an OD value of 0.1−0.2 at 625 nm. The bacterial inhibition percentage
onto an aluminum foil, air-dried, and sputter-coated with a carbon film
was determined at different time points (1, 2, 6, 12, 24, 48, 72, and 96
with a thickness of 10 nm before measurement. The crystalline
h, respectively). For comparison, PLGA/AMX nanofibers were also
structure of LAP, AMX, and LAP/AMX nanodisks was characterized
tested under similar conditions.
by a Rigaku D/max-2550 PC X-ray diffraction (XRD) system (Rigaku
Another antibacterial activity testing method based on solid
Co., Tokyo, Japan) using Cu Kα radiation with a wavelength of 1.54 Å
medium was also used in this study.Briefly, agar (1.5 g) was
at 40 kV and 200 mA. The scan was performed from 5° to 60° (2θ).
added into 100 mL liquid medium and autoclaved. Then, the agar
The plane spacing of different diffraction planes (dhkl) can be
medium was poured onto Petri dishes and air-dried. The PLGA,
calculated from the Bragg's Law:
PLGA/LAP, PLGA/AMX, and PLGA/LAP/AMX nanofibrous mats
were cut into small pieces with a diameter of about 1 cm and the same
weight. After that, the solid agar medium plates were seeded with 100
μL of S. aureus suspension and covered with PLGA, PLGA/LAP,
where λ is the wavelength of the copper anode source (λ = 1.54 Å) and
PLGA/AMX, and PLGA/LAP/AMX nanofibrous mats, respectively,
θ stands for the diffraction angle of each indexed diffraction plane. The
for an antibacterial activity assay. In another method, the solid agar
morphology of PLGA, PLGA/LAP, PLGA/AMX, and PLGA/LAP/
medium plates were first covered with the PLGA, PLGA/LAP, PLGA/
AMX nanofibers was observed using scanning electron microscopy
AMX, and PLGA/LAP/AMX nanofibrous mats, respectively, and the
(SEM) (JEOL JSM-5600LV, Japan) with an accelerating voltage of 10
fibrous mats were removed after a 4 h incubation. Then, 100 μL S.
kV. Before measurement, each sample was sputter-coated with a 10
aureus suspension was seeded onto each sample-treated solid medium.
nm-thick gold film. Fiber diameter was measured using Image J 1.40 G
All of these agar plates were incubated at 37 °C for the given time
software (At least 100
period. The bacterial inhibition zones were visually observed to test
nanofibers from different SEM images for each sample were randomly
the samples' antibacterial activity.
selected and analyzed. Water contact angle test was used to evaluate
Cytocompatibility Evaluation. For cytocompatibility evaluation,
the surface hydrophilicity of the PLGA/AMX and PLGA/LAP/AMX
PLGA and PLGA/LAP/AMX nanofibers were prepared on coverslips
fibrous mats as reported in our previous study.In brief, a pendant
with a diameter of 14 mm. Then, these mats were fixed in 24-well
droplet of water with 1 μL drop size was dropped onto the surface of
plates with stainless steel rings and sterilized with 75% alcohol for 2 h.
each sample at the randomly selected area at ambient temperature and
After that, all wells with samples were washed 3 times with PBS
humidity. The contact angle was measured three times for each sample
solution to remove the residual alcohol. Finally, 1 mL of complete
using a contact angle goniometer (DSA-30, Kruss, Germany) when the
DMEM was added to individual wells to incubate at 37 °C overnight.
droplet was stable.
PIEC cells were seeded at a density of 1.5 × 104 cells/well for MTT
In Vitro Drug Release. The in vitro release kinetics of AMX from
assay and 2 × 104 cells/well for SEM morphology observation,
LAP/AMX nanodisks, PLGA/AMX nanofibers, and PLGA/AMX/
respectively. Coverslips without nanofibers and tissue culture plates
LAP nanofibers was studied using UV−vis spectroscopy. Briefly, LAP/
(TCPs) were used as controls.
AMX nanodisks (6 mg) were dispersed into 1 mL of phosphate
After cell seeding for 8 h or 3 days, unattached cells were washed
buffered saline (PBS) solution (pH = 7.4) and placed in a dialysis bag
out with PBS solution and MTT solution (40 μL) diluted with fresh
with a molecular weight cutoff of 10 000 and then dialyzed against 2
medium (360 μL) was added to each well. After being incubated at 37
mL of PBS solution in a sample vial. For the nanofibers, 24 mg of
°C for 4 h, 400 μL of DMSO was added to dissolve the purple MTT
PLGA/AMX or PLGA/AMX/LAP nanofibers was dipped into a
formazan crystal. Then, 100 μL of the dissolved formazan solution of
sample vial containing 3 mL of PBS solution. All these samples were in
each sample was transferred into individual wells of a 96-well plate to
triplicate and were incubated in a vapor-bathing constant temperature
test the OD value at 570 nm using a microplate reader (MK3,
vibrator at 37 °C for different time periods. At each time interval, 1 mL
Thermo, USA). Mean and standard deviation for the triplicate wells
of PBS solution was taken out from each vial and an equal volume of
for each sample were reported.
dx.doi.org/10.1021/am302130b ACS Appl. Mater. Interfaces 2012, 4, 6393−6401
ACS Applied Materials & Interfaces
After being cultured for 8 h or 3 days, samples were rinsed 3 times
aggregation of the LAP nanodisks at higher concentrations,
with PBS solution and then fixed with 2.5 wt % glutaraldehyde at 4 °C
leading to decreased accessibility of the drug molecules to the
for 2 h. After that, the samples were dehydrated through a series of
interlayer space of LAP. We also note that the optimized
gradient ethanol solutions of 30%, 50%, 70%, 80%, 90%, 95%, and
loading efficiency of 9.76 ± 0.57% may not be the highest
100% and air-dried overnight. The morphology of cells was observed
loading efficiency; further adjusting the concentrations of LAP,
by SEM (JEOL JSM-5600LV) with an accelerating voltage of 10 kV,and the samples were sputter coated with a 10 nm thick gold film
AMX, and the solution pH is necessary to achieve the
maximum loading efficiency.
Statistical Analysis. One-way ANOVA statistical analysis was
The successful encapsulation of AMX within LAP was
performed to compare the cytocompatibility of cells cultured onto
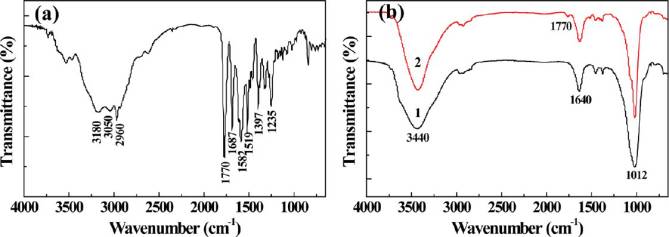
confirmed using FTIR spectroscopy (Figure In the FTIR
different materials and to compare the bacterial inhibition effect of the
spectrum of AMX powder (Figure the typical absorption
tested materials with different AMX concentrations in liquid medium.
bands at 1687, 1519, and 1235 cm−1 can be assigned to the
0.05 was selected as the significance level, and the data were indicated
amide I, amide II, and amide III bond of AMX, respectively.
with (*) for p < 0.05, (**) for p < 0.01, and (***) for p < 0.001,
The weak peaks at 1770 and 1397 cm−1 may be attributed to
the vibration of carbonyl group and carboxyl group of the
RESULTS AND DISCUSSION
AMX, respectively.The peaks at 3180 and 3050 cm−1 areassigned to the stretching vibration of free amino group in the
Loading of AMX within LAP Nanodisks. Different from
AMX structure. The peak of 2960 cm−1 can be assigned to the
our previous study related to the use of the lumen of HNTs for
stretching vibration for −CH−, −CH −, or −CH
drug the interlayer space of LAP nanodisks was
structure. In the spectrum of LAP and LAP/AMX nanodisks
used to encapsulate a model drug AMX. LAP nanodisks have a
(Figure the moderate peak at 1640 cm−1 may be caused by
two-dimensional structure with six octahedral magnesium ions
the moisture from the atmosphere. The strong peak located at
sandwiched between two layers of four tetrahedral silicon
1012 cm−1 can be assigned to the −Si−O− stretching vibration
atoms,and the interlayer space of LAP nanodisks has been
of LAP naondisks,and the broad peak at 3440 cm−1 may be
proven to be used as a reservoir for drug encapsulation.
due to the bending vibration of −OH in the LAP structure. By
The loading amount and the loading efficiency of AMX
comparing the spectrum of LAP with that of LAP/AMX, a new
within LAP were determined using the standard concen-
peak emerged at 1770 cm−1 in the spectrum of LAP/AMX
tration−absorbance (at 230 nm) calibration curve of AMX in
suggesting the successful encapsulation of AMX into LAP. Due
water and was optimized by changing the concentration of LAP
to the quite low amount of AMX encapsulated within the LAP,
and AMX, respectively. As shown in Figure an optimized
it is difficult to observe some other distinctive peaks of AMXdrug.
The morphology of LAP nanodisks before and after
encapsulation of AMX was observed with FESEM (Figure S1,It is clear that the disk-shaped LAPdoes not significantly change after the encapsulation of AMX,indicating the successful intercalation of drug molecules withinthe LAP interlayer space. The somewhat aggregated particlesshown in the FESEM images for both samples are presumablydue to the sample preparation method, which includes the air-drying process. As reported in our previous study, the air-dryingof the aqueous suspension of the samples before measurementmay lead to a partial aggregation or interconnection of the
Figure 1. AMX loading efficiency as a function of AMX concentration
The LAP nanodisks are able to form a stable colloidal layered
under different LAP concentrations.
structure in aqueous solution, which facilitates drug encapsu-lation.The encapsulation of drug within the LAP interlayer
loading efficiency of 9.76 ± 0.57% could be achieved when the
space may result in a change in the interlayer distance,and
AMX and LAP concentration was 2 and 3 mg/mL, respectively.
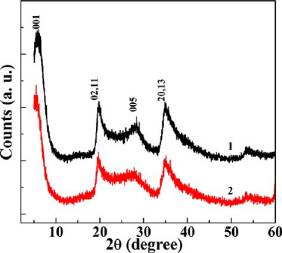
this can be determined by XRD technology. The XRD patterns
It is worthwhile to note that the loading efficiency decreases
of the LAP nanodisks before and after AMX encapsulation were
with the LAP concentration, which is likely due to the prone
compared, and the data are shown in Figure and Table
Figure 2. FTIR spectra of pure AMX (a) and LAP before (Curve 1) and after (Curve 2) AMX loading (b).
dx.doi.org/10.1021/am302130b ACS Appl. Mater. Interfaces 2012, 4, 6393−6401

ACS Applied Materials & Interfaces
Figure 3. XRD patterns of LAP nanodisks before (Curve 1) and after(Curve 2) AMX loading.
Table 1. Diffraction Angle and Plane Spacing Data of LAPand LAP/AMX from XRD Analysis
2θ peak position (o)
plane spacing (d, Å)
diffraction plane (hkl)
Obviously, most of the diffraction planes at their correspondingdiffraction angles do not change, suggesting that LAP is able tomaintain its crystalline structure after AMX The diffraction angle of (001) plane shifted from 6.06° to5.50°, and the plane spacing was larger (from 14.76 to 16.26 Å)after AMX encapsulation. This is likely due to the fact that theAMX molecules are intercalated along the 001 plane. Besides,when compared with AMX powder (Figure S2, no diffraction peaks of AMX can be detected inLAP/AMX nanodisks, which is presumably ascribed to the factthat the amount of the incorporated drug is too small to bedetectable by the XRD technique. The XRD data suggested
Figure 4. SEM micrographs and diameter distribution histograms of
that the incorporation of AMX within LAP is primarily via the
(a) PLGA, (b) PLGA/LAP (5 wt % LAP relative to PLGA), (c)
drug intercalation within the LAP interlayer space. It is also
PLGA/AMX (0.5 wt % AMX relative to PLGA), and (d) PLGA/
possible that a small portion of AMX can be adsorbed onto the
AMX/LAP (5 wt % LAP relative to PLGA) nanofibers.
LAP surface via hydrogen bonding or other weak forces.
Formation of PLGA/LAP/AMX Nanofibers. The AMX-
(929 nm, Figure presumably due to the increase of the
loaded LAP was then incorporated within PLGA nanofibers
solution conductivity, which was caused by the introduction of
(with 0.5% AMX relative to PLGA) via electrospinning to form
an anionic or a cationic species in the electrospinning solution.
PLGA/LAP/AMX nanofibers (Scheme As controls, PLGA,
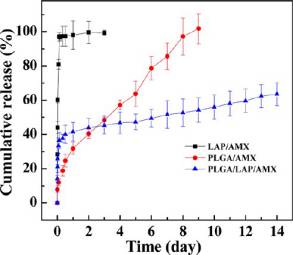
Release of AMX from PLGA/LAP/AMX Composite
PLGA/LAP (with 5% LAP relative to PLGA), and PLGA/
Nanofibers. The in vitro drug release property of PLGA/
AMX (0.5% AMX relative to PLGA) nanofibers were prepared
LAP/AMX composite nanofibers was investigated by exposure
in the same manner. The successful incorporation of LAP
of the fibrous mats in PBS solution (pH = 7.4) at 37 °C. The
within PLGA nanofibers has been confirmed by thermogravi-
AMX release profile was compared with those from LAP/AMX
metric analysis, transmission electron microscopy, porosity
nanodisks and PLGA/AMX nanofibers with similar drug
measurement, mechanical testing, and contact angle measure-
content (Figure It is clear that the AMX in the LAP/
ment (see also Figure S3, in our
AMX nanodisks has a burst release profile and about 97% of the
previous work.Here, in this study, SEM was used to
encapsulated AMX can be released from LAP within 3 h. In
characterize the morphology of the formed electrospun
sharp contrast, the AMX release rate from PLGA/AMX and
nanofibers with different compositions (Figure Similar to
PLGA/LAP/AMX nanofibers was significantly reduced and
our previous studies related to the formation of PLGA/HNTs
showed a sustained manner. The release of AMX from PLGA/
composite nanofibers,we were able to form electrospun
AMX nanofibers showed a moderate rate on the first day, and
PLGA/LAP composite nanofibers with a smooth and uniform
31.8% of the AMX was released. Then, the release speed was
fibrous morphology even after AMX encapsulation, similar to
slowed down, and approximately 100% drug release was
the pure PLGA and PLGA/drug nanofibers. The diameters of
achieved on the ninth day. The slower AMX release rate from
PLGA/LAP (550 nm, Figure PLGA/AMX (842 nm,
PLGA/AMX nanofibers than that from LAP/AMX nanodisks is
Figure and PLGA/LAP/AMX (591 nm, Figure
likely due to the effective hydrogen bonding and electrostatic
nanofibers are smaller than that of pure PLGA nanofibers
interactions between the hydroxyl, amine, and carboxyl groups
dx.doi.org/10.1021/am302130b ACS Appl. Mater. Interfaces 2012, 4, 6393−6401

ACS Applied Materials & Interfaces
Figure 5. In vitro release of AMX from LAP/AMX nanodisks, PLGA/
Figure 6. Growth inhibition of S. aureus after treatment with AMX
AMX nanofibers, and PLGA/LAP/AMX nanofibers.
powder (1), LAP/AMX (2), PLGA/AMX nanofibers (3), and PLGA/LAP/AMX nanofibers (4) with different AMX concentrations for 24 hat 37 °C in liquid medium.
of AMX and the carboxyl residues of PLGA polymer. The drugrelease profile of the PLGA/LAP/AMX composite nanofibersfollows a biphasic pattern characterized by an initial fast release
antibacterial activity of LAP/AMX nanodisks decreased with
and a followed sustained release phase after 12 h. 40.2% of the
the drug concentration. This is likely due to the increased
AMX was released within the first 12 h, and a sustained release
concentration of LAP in the liquid medium, which can absorb
with a relatively low rate remained; 63.5% AMX was released
AMX molecules back to compromise the drug efficacy to some
on the 14th day. Since PLGA is biodegradable and LAP has a
extent. The bacterial inhibition of PLGA/AMX and PLGA/
swelling ability in PBS solution,it is believed that all of the
LAP/AMX nanofibers increased with the drug concentration
encapsulated AMX can be released with time. The release rate
and was higher than 90% at each concentration. There was no
of the PLGA/LAP/AMX nanofibers was faster than that of the
statistically significant difference between pure AMX powder
PLGA/AMX nanofibers in the first 2 days and then showed a
and PLGA/LAP/AMX nanofibers at the same AMX concen-
slower and sustained release rate.
tration in terms of the bacterial inhibition efficacy (p > 0.05),
The burst release of the LAP/AMX nanodisks may be due to
suggesting that the PLGA/LAP/AMX composite nanofibers
the swelling behavior of the colloidal LAP. After contact with
have a comparable bacterial inhibition efficacy with that of the
the PBS solution, the LAP nanodisks swell and the
pure AMX powder. In contrast, PLGA and PLGA/LAP
incorporated AMX molecules can be quickly released. The
nanofibers without AMX encapsulation did not have any
initial fast release of the PLGA/LAP/AMX nanofibers may be
antibacterial efficacy, similar to the untreated negative control.
due to the inevitable release of AMX from LAP/AMX
The correlation of the antibacterial activity of PLGA/LAP/
nanodisks when they were mixed with PLGA solution before
AMX nanofibers as a function of AMX release time was also
electrospinning. After the formation of PLGA/LAP/AMX
investigated (Figure S4, Apparently,
nanofibers, the partially released AMX can be attached onto
at all the release time points (1, 2, 6, 12, 24, 48, 72, and 96 h,
the nanofiber surface or dispersed throughout the polymer
respectively), the released AMX from PLGA/LAP/AMX
matrix in a matrix-type structure, thereby causing an initial burst
nanofibers can effectively inhibit the bacterial growth, similar
release. It is interesting to note that the initial burst release of
to that from PLGA/AMX nanofibers.
PLGA/LAP/AMX nanofibers is higher than that of PLGA/
The bacterial inhibition activity of the PLGA/LAP/AMX
AMX. This can be explained as follows: The diameter of
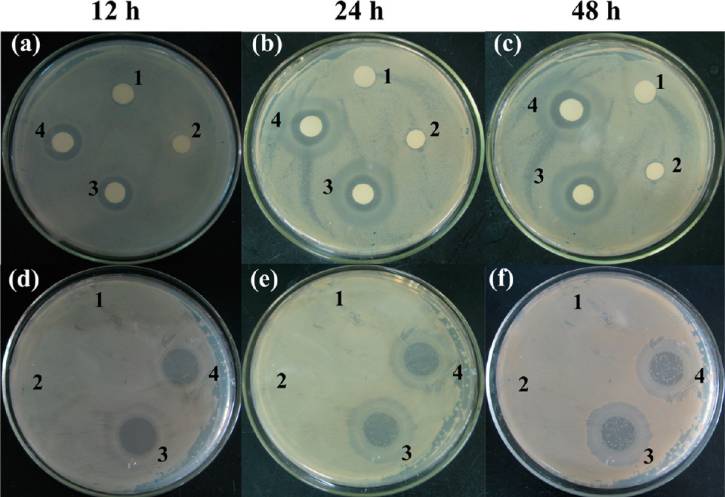
composite nanofibers was also tested onto solid medium.
PLGA/LAP/AMX nanofibers is apparently smaller than that of
Figure shows the digital photos of the antibacterial circles on
PLGA/AMX (as shown in Figure which shortened the drug
agar plates at different culture times. PLGA (1), PLGA/LAP
diffusion distance between the PLGA fiber matrix to the release
(2), PLGA/AMX (3), and PLGA/LAP/AMX (4) nanofibers
medium. The followed slow release speed of the PLGA/LAP/
were pasted onto the agar plate for bacteria inhibition (Figure
AMX relative to the PLGA/AMX nanofibers is easily
−c). Obviously, both the PLGA/AMX and PLGA/LAP/
understandable due to the coexistence of two types of drug-
AMX nanofibers were able to effectively inhibit bacterial
carriers, namely, reservoir-type and matrix-type (Scheme
growth, and the zones of inhibition for PLGA/LAP/AMX and
The drug should first come out from the reservoir of LAP and
PLGA/AMX are basically similar in size after a 12, 24, and 48 h
then from the polymer matrix, which provides an additional
culture, implying that the PLGA/LAP/AMX nanofibers has a
barrier for the drug
good bacterial inhibition efficacy under the studied conditions.
In Vitro Antibacterial Activity Assay of Nanofibers. For
The bacterial inhibition efficacy of the PLGA/LAP/AMX
development of novel, effective drug delivery systems, it is
nanofibers was further confirmed by removing the nanofibrous
important to maintain the activity of the drug after
mats from the agar plate after a 4 h release of AMX, followed by
encapsulation within the composite PLGA/LAP nanofibers.
bacterial seeding (Figure −f). Similar to the above method,
We next explored the in vitro antibacterial activity of the AMX-
PLGA/AMX and PLGA/LAP/AMX nanofibers were able to
loaded nanofibers using S. aureus as a model bacterium both in
effectively inhibit the bacterial growth. In contrast, PLGA and
liquid and on solid medium. Figure shows the bacterial
PLGA/LAP nanofibers without AMX encapsulation did not
inhibition assay results of AMX powder, LAP/AMX nanodisks,
inhibit the bacterial growth in both cases, implying that the
PLGA/AMX nanofibers, and PLGA/LAP/AMX nanofibers in
bacterial inhibition effect is solely related to the encapsulated
liquid medium with different AMX concentrations (10, 20, and
AMX drug. It should be noted that, for solid medium testing,
30 μg/mL, respectively). The AMX powder was able to inhibit
we just tested all the nanofibrous samples because it was
the bacterial growth at each studied concentration, while the
difficult to uniformly lay down the solid powder samples of free
dx.doi.org/10.1021/am302130b ACS Appl. Mater. Interfaces 2012, 4, 6393−6401
ACS Applied Materials & Interfaces
Figure 7. Inhibition of S. aureus cultured on agar plate incubated at 37 °C at 12, 24, and 48 h. In panels a−c, nanofibers were pasted onto the agarplate for the whole culture time period. In panels d−f, nanofibers were removed after a 4 h release of AMX onto the agar plate. 1−4 representsPLGA, PLGA/LAP, PLGA/AMX, and PLGA/LAP/AMX nanofibers, respectively.
AMX and LAP/AMX for effective comparison. Taken togetherwith the data obtained in liquid medium, we can conclude thatthe developed PLGA/LAP/AMX composite nanofibers are ableto inhibit the growth of a model bacterium, and the loading ofAMX within the composite nanofibers does not compromisethe inherent antibacterial activity of the drug. It should benoted that, although the PLGA/LAP/AMX and PLGA/AMXnanofibers showed different drug release patterns, theantimicrobial activity was almost the same. However, themajor advantage of the PLGA/LAP/AMX nanofibers is that thesustained release of the drug from the fibers is very importantfor certain biomedical applications requiring the drug to have a
Figure 8. MTT viability assay of PIEC cells seeded on the TCPs
long-term therapeutic efficacy. Besides, the incorporation of
(control), cover slips (control), PLGA nanofibers, and PLGA/LAP/
LAP within PLGA nanofibers can significantly enhance the
AMX nanofibers (mean ± SD, n = 3, *p < 0.05).
mechanical property of PLGA nanofibers.
Cytocompatibility of the PLGA/LAP/AMX Composite
the time point of 8 h and 3 days does not impact the cell
Nanofibers. To further validate the potential biomedical
applications of the developed PLGA/LAP/AMX composite
The comparison of the cytocompatibility of PLGA/LAP/
nanofibers, we next tested the cytocompatibility of the fibers via
AMX composite nanofibers with pure PLGA nanofibers was
MTT assay in comparison with pure PLGA nanofibers with
also validated via the cell morphology observation. The
proven biocompatibility.The viability of PIEC cells
morphologies of PIEC cells cultured onto PLGA and PLGA/
cultured onto both PLGA and PLGA/LAP/AMX nanofibers
LAP/AMX nanofibers after an 8 h and 3 day culture are shownin Figure Obviously, cells are able to attach onto both
after 8 h and 3 days is shown in Figure No statistically
nanofibrous scaffolds after an 8 h culture, and after 3 days, the
significant difference can be found among each sample after an
cells cultured onto both nanofibrous scaffolds display a
8 h culture, indicating that both PLGA and PLGA/LAP/AMX
phenotypic shape, indicating that the cells can penetrate and
nanofibers display similar adhesion viability, in comparison with
migrate within the scaffolds in a manner similar to native
the coverslips and TCPs. On day 3, the proliferation viability of
extracellular matrix. These cell morphology observation data
PIEC cells cultured onto both PLGA and PLGA/LAP/AMX
corroborate the results of the MTT assay.
nanofibers is significantly higher than those onto coverslips andTCPs (p < 0.05), and no significant difference exists between
the PLGA and PLGA/LAP/AMX nanofibers (p > 0.05). This
In summary, we developed a facile approach to encapsulating
implies that both PLGA and PLGA/LAP/AMX nanofibers have
an antibiotic drug AMX within PLGA/LAP composite
an excellent cytocompatibility, and the incorporation of LAP/
nanofibers for biomedical applications. The AMX-loaded LAP
AMX nanodisks does not compromise the cytocompatibility of
nanodisks with an optimized loading efficiency of 9.76 ± 0.57%
PLGA nanofibers. This also indicates that the released AMX at
were able to be incorporated within PLGA nanofibers without
dx.doi.org/10.1021/am302130b ACS Appl. Mater. Interfaces 2012, 4, 6393−6401
ACS Applied Materials & Interfaces
Figure 9. SEM images of PIEC cells cultured onto PLGA (a, c) and PLGA/LAP/AMX (b, d) nanofibers after an 8 h (a, b) and 3 day (c, d) culture.
significantly changing the PLGA fibrous morphology. With the
Donghua University Doctorate Dissertation of Excellence
coexistence of both the reservoir-type of LAP interlayer space
(BC201107). M.Z. thanks the National Natural Science
and the matrix-type of PLGA nanofibers, the release profile of
Foundation of China (50925312) for support.
AMX was able to be significantly improved with a biphasic andsustained manner. Furthermore, PLGA/LAP/AMX nanofibers
display effective antibacterial activity and noncompromisedcytocompatibility in comparison with pure PLGA nanofibers.
(1) Greiner, A.; Wendorff, J. H. Angew. Chem., Int. Ed. 2007, 46,
Such preparation of PLGA/LAP/AMX composite nanofibers
(2) Li, D.; Xia, Y. Adv. Mater. 2004, 16, 1151−1170.
may be able to be extended for other drug encapsulation and
(3) Wang, S.; Cao, X.; Shen, M.; Guo, R.; Bányai, I.; Shi, X. Colloids
release for various biomedical applications in the fields of
Surf., B: Biointerfaces 2012, 89, 254−264.
tissues engineering and pharmaceutical science.
(4) Lu, X.; Wang, C.; Wei, Y. Small 2009, 5, 2349−2370.
(5) Qi, R.; Guo, R.; Shen, M.; Cao, X.; Zhang, L.; Xu, J.; Yu, J.; Shi,
ASSOCIATED CONTENT
X. J. Mater. Chem. 2010, 20, 10622−10629.
*S Supporting Information
(6) Moreno, I.; González-González, V.; Romero-García, J. Eur. Polym.
Additional materials characterization data and bacterial
J. 2011, 47, 1264−1272.
inhibition data. This information is available free of charge
(7) Peining, Z.; Nair, A. S.; Shengjie, P.; Shengyuan, Y.; Ramakrishna,
via the Internet at
S. ACS Appl. Mater. Interfaces 2012, 4, 581−585.
(8) Zhao, Y.; Wang, S.; Guo, Q.; Shen, M.; Shi, X. J. Appl. Polym. Sci.
2012, DOI: 10.1002/APP.38054.
AUTHOR INFORMATION
(9) Parajuli, D. C.; Bajgai, M. P.; Ko, J. A.; Kang, H. K.; Khil, M. S.;
Corresponding Author
Kim, H. Y. ACS Appl. Mater. Interfaces 2009, 1, 750−757.
(10) Kenawy, E. R.; Bowlin, G. L.; Mansfield, K.; Layman, J.;
Author Contributions
Simpson, D. G.; Sanders, E. H.; Wnek, G. E. J. Controlled Release 2002,81, 57−64.
S.W. and F.Z. equally contributed to this work.
(11) Shen, X.; Yu, D.; Zhu, L.; Branford-White, C.; White, K.;
Chatterton, N. P. Int. J. Pharm. 2011, 408, 200−207.
The authors declare no competing financial interest.
(12) Yoo, H. S.; Kim, T. G.; Park, T. G. Adv. Drug Delivery Rev. 2009,
61, 1033−1042.
(13) Wang, C.; Yan, K. W.; Lin, Y. D.; Hsieh, P. C. H. Macromolecules
2010, 43, 6389−6397.
This research is financially supported by the Program for New
(14) Bölgen, N.; Vargel, I.; Korkusuz, P.; Menceloölu, Y. Z.; Pişkin,
Century Excellent Talents in University, State Education
E. J. Biomed. Mater. Res., Part B 2007, 81, 530−543.
Ministry, the High-Tech Research and Development Program
(15) Xu, X.; Chen, X.; Ma, P.; Wang, X.; Jing, X. Eur. J. Pharm.
of China (2012AA030309), ‘‘111 Project'', B07024, and the
Biopharm. 2008, 70, 165−170.
Fundamental Research Funds for the Central Universities (for
(16) Moghe, A. K.; Professor, B. S. G. Polym. Rev. 2008, 48, 353−
M.S. and X.S.). X.S. gratefully acknowledges the Fundação para
a Ciência e a Tecnologia (FCT) and Santander bank for the
(17) Qi, H.; Hu, P.; Xu, J.; Wang, A. Biomacromolecules 2006, 7,
Chair in Nanotechnology. S.W. thanks the Innovation Funds of
dx.doi.org/10.1021/am302130b ACS Appl. Mater. Interfaces 2012, 4, 6393−6401
ACS Applied Materials & Interfaces
(18) Jung, H.; Kim, H. M.; Choy, Y. B.; Hwang, S. J.; Choy, J. H. Int.
J. Pharm. 2008, 349, 283−290.
(19) Jung, H.; Kim, H. M.; Choy, Y. B.; Hwang, S. J.; Choy, J. H.
Appl. Clay Sci. 2008, 40, 99−107.
(20) Viseras, C.; Cerezo, P.; Sanchez, R.; Salcedo, I.; Aguzzi, C. Appl.
Clay Sci. 2010, 48, 291−295.
(21) Liu, F.; Guo, R.; Shen, M.; Cao, X.; Mo, X.; Wang, S.; Shi, X.
Soft Mater. 2010, 8, 239−253.
(22) Liu, F.; Guo, R.; Shen, M.; Wang, S. H.; Shi, X. Macromol.
Mater. Eng. 2009, 294, 666−672.
(23) Wang, S.; Castro, R.; An, X.; Song, C.; Luo, Y.; Shen, M.;
Tomás, H.; Zhu, M.; Shi, X. J. Mater. Chem. 2012, 22, 23357−23367.
(24) Dai, J.; Bruening, M. L. Nano Lett. 2002, 2, 497−501.
(25) Zhang, Y.; Peng, H.; Huang, W.; Zhou, Y.; Zhang, X.; Yan, D. J.
Phys. Chem. C 2008, 112, 2330−2336.
(26) Kim, K.; Luu, Y. K.; Chang, C.; Fang, D.; Hsiao, B. S.; Chu, B.;
Hadjiargyrou, M. J. Controlled Release 2004, 98, 47−56.
(27) Tai, Z.; Ma, H.; Liu, B.; Yan, X.; Xue, Q. Colloids Surf., B:
Biointerfaces 2011, 89, 147−151.
(28) Boyle, V. J.; Fancher, M. E.; Ross, R. W., Jr. Antimicrob. Agents
Chemother. 1973, 3, 418−424.
(29) Herrera, N. N.; Letoffe, J. M.; Putaux, J. L.; David, L.; Bourgeat-
Lami, E. Langmuir 2004, 20, 1564−1571.
(30) Bisson-Boutelliez, C.; Fontanay, S.; Finance, C.; Kedzierewicz,
F. AAPS PharmSciTech 2010, 11, 574−581.
(31) Shi, X.; Briseno, A. L.; Sanedrin, R. J.; Zhou, F. Macromolecules
2003, 36, 4093−4098.
(32) Takahashi, T.; Yamada, Y.; Kataoka, K.; Nagasaki, Y. J.
Controlled Release 2005, 107, 408−416.
(33) Mercier, L.; Pinnavaia, T. J. Microporous Mesoporous Mater.
1998, 20, 101−106.
(34) Park, M.; Shim, I. K.; Jung, E. Y.; Choy, J. H. J. Phys. Chem.
Solids 2004, 65, 499−501.
(35) Qi, R.; Cao, X.; Shen, M.; Guo, R.; Yu, J.; Shi, X. J. Biomater. Sci.,
Polym. Ed. 2012, 1, 299−313.
(36) Qi, R.; Shen, M.; Cao, X.; Guo, R.; Tian, X.; Yu, J.; Shi, X.
Analyst 2011, 136, 2897−2903.
(37) Chen, M.; Gao, S.; Dong, M.; Song, J.; Yang, C.; Howard, K. A.;
Kjems, J.; Besenbacher, F. ACS Nano 2012, 6, 4835−4844.
dx.doi.org/10.1021/am302130b ACS Appl. Mater. Interfaces 2012, 4, 6393−6401
Source: http://cqm.uma.pt/CQM12/12-41_ACS.pdf
Rapid detection of genetically modified organisms on a continuous-flow polymerase chain reaction microfluidics
Analytical Biochemistry 385 (2009) 42–49 Contents lists available at Analytical Biochemistry Rapid detection of genetically modified organisms on a continuous-flowpolymerase chain reaction microfluidics Yuyuan Li, Da Xing *, Chunsun Zhang MOE Key Laboratory of Laser Life Science and Institute of Laser Life Science, South China Normal University, No. 55, Zhongshan Avenue West, Tianhe District,Guangzhou 510631, People's Republic of China
Microsoft word - 5.22-27.doc
j. innov.dev.strategy. 2(2): 22-27 (July 2008) ANTIMICROBIAL SUSCEPTIBILITY OF Salmonella SEROVARS ISOLATED FROM BLOOD M. J. ISLAM1, K. K. DAS2, N. SHARMIN3, M. N. HASAN4 AND A. K. AZAD5 1Lecturer, Department of Pharmacy, University of Development Alternative, Dhanmondi, Dhaka, 2, 3 & 4Lecturer, Department of Biotechnology and Genetic Engineering, 5Assistant professor, Department of Pharmacy, University of Development Alternative, Dhanmondi, Dhaka, Bangladesh.