Techpart.co.uk




Pulmonary Delivery
Fresh Air
echnology P
Images: The T
In the highly competitive world of drug discovery and development,
By Iain McDerment at The Technology
it is easy to focus on the early stages of identifying a candidate
molecule or the fi rst critical rounds of clinical trials. However, in addition to selecting an attractive druggable target, the method of delivery is an equally important fi eld of research, since it can infl uence both product effi cacy and patient behaviour
Drug delivery is big business, and
can have a number of additional
Currently, the major solutions
estimates of the global market
consequences to the patient.
for non-invasive drug delivery
for this area of technology put
The route chosen can potentially
are focused on the oral, inhaled,
revenues at $122.3 billion in
contribute to product line
transdermal, ocular and nasal
extensions, provide a more patient-
routes, and make use of a
expected rise to
friendly dosage form, and play
broad selection of devices and
$198.4 billion in
an important role in extending a
technologies (2). Novel delivery
2017 (1). Clearly,
medicine's lifecycle. Importantly,
technology is essential for
Inhaled drug delivery
this is a market
innovative drug delivery
optimising drug efficacy,
technology has the capacity
reducing risk profiles and
to improve the medication's
potentially aiding patient
User-friendly technology
bioavailability, as well as its
compliance. The development
Over-the-counter market
safety profi le (1).
of new devices also opens up
Innovations in Pharmaceutical Technology Issue 52

the interesting possibility
of introducing products
to specialised markets –
paediatrics, for example. For
this reason, some companies
are now turning their attention
to innovation in the area of
drug delivery devices.
Targeting the Lungs
Pulmonary delivery has been
attracting interest recently due
to the unique structure of the
lung, which provides a large
surface area, an exceedingly
thin absorptive mucosal
membrane and an excellent
blood supply (3). These
physiological parameters
lend themselves very well
as a mode of drug delivery,
providing the benefits of a
rapid onset of action, high
local concentration and efficient
Excessive degradation of a drug in
the evolution of inhalation
systemic delivery. The latter is of
the liver (first-pass metabolism) is
devices, including pressurised
special interest for drugs that may
also avoided when administered
metered dose inhalers (pMDIs),
otherwise have poor bioavailability
via inhalation.
dry powder inhalers and liquid
when administered orally (4).
aerosol devices. Many of the
latest developments have been
In the treatment of pulmonary
completed with collaborative
illnesses such as asthma and
The pulmonary route is a
efforts, benefiting from a large
chronic obstructive pulmonary
powerful means of enhancing
pool of expertise. This article
disease (COPD), inhaled delivery
the administration of certain
looks at some of the issues
deposits the drug directly
drugs, offering potentially
that have been faced by those
to affected areas at high
improved bioavailability, lower
developing inhalation devices.
concentrations. Additionally,
doses and reduced side-effects
this route means that the overall
over traditional solid oral dosage
amount of medicine needed to
forms. However, developing
A key problem commonly seen in
be given to the patient is reduced:
suitable devices can be challenging
inhalation devices is that they lack
one 4mg tablet of salbutamol,
– the size of the inhaled particle,
any features capable of informing
for example, is the equivalent
for example, is especially important:
the user just how many doses
40 inhaled metered doses (3).
too small and the particle will
of a drug remain in an inhaler.
This can minimise systemic
be exhaled, yet overly large
With a typical pMDI, the amount
side-effects, as well as provide
particles can adversely affect the
of available drug must be guessed,
a faster drug response.
throat and larynx (3). Another vital
with patients typically having
component of inhaled drug design
to shake the device and simply
From a pharmacological
is ensuring that the device itself is
estimate how much is left – but this
perspective, inhaling a product
user-friendly: capable of delivering
can be hard to do accurately, given
avoids many of the problems
the medication efficiently, while
the relative weights of the drug
associated with swallowing,
providing the patient with
including poor solubility, low
something pleasant to use.
bioavailability, gut irritability,
A collaboration between
unwanted metabolites, food
A wide variety of drug delivery
AstraZeneca and The Technology
effects and dosing variability (3).
technologies are used to drive
Partnership (TTP) aimed to address
Innovations in Pharmaceutical Technology Issue 52
to meet requirements while
frequencies against a body
ensuring the safe administration
of fluid, generating a stream of
of an antiviral drug.
precisely controlled droplets.
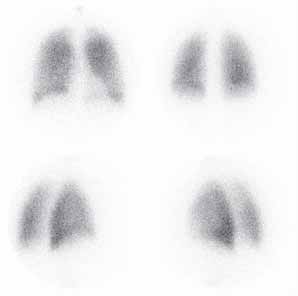
Initial in vivo work shows that
Launched by Daiichi Sankyo,
83% of a delivered aerosol
Japan's second largest
appears on a lung scintigraph (6),
pharmaceutical company,
and in vitro measurements
TwinCaps was marketed for the
from human insulin (Velosulin®)
delivery of its antiviral drug,
and bovine serum albumin
Invair®, and outsold Relenza and
suggest TouchSpray is capable
Tamiflu within the first six months.
of aerosolising these with
During in vitro tests, TwinCaps
mean recovery rates in excess
was shown to deliver 95% of
the nominal dose at 20 litres per
minute of airflow, and 100% at 25
litres per minute and above (5).
TouchSpray technology
ultimately led to a collaborative
Making the Perfect Droplet
effort between TTP and PARI,
this issue, using a highly reliable
The dry powder delivery
culminating in the global launch
counter in the form of a dial with
devices mentioned thus far
of the PARI eFlow® electronic
Scintigraph showing deep lung penetration
a colour display that sits atop the
are innovative and useful in
nebuliser – an inhaler that
of aerosols using the
device, with a clear dose count
diverse ways, but these forms
cuts patient treatment time by
TouchSpray spray
from 120 down to zero. The dial's
are perhaps less than optimal
50%. The device enables liquid
engine (diethylene triamine pentaacetic
display provides users with a
for the delivery of protein- and
medication to be absorbed
acid solution 3mg/mL,
quantifiable number of doses
peptide-based drugs. For this,
through the lungs with as
D[4,3] < 1μm)
remaining in their inhaler, offering
a liquid-based drug is superior,
much as 90% efficacy.
both patient value and significant
yet making liquid amenable
to the pulmonary route has its
This system uses a chamberless,
own difficulties. Establishing an
planar nozzle plate driven by
optimal droplet size for inhalation
a piezoelectric actuator, and
In order to meet tight
devices dispensing a liquid
the inertial transfer mechanism
commercial timelines driven
drug is imperative: for efficient
generates a highly defined
by the Japanese government's
deep lung penetration, a device
aerosol of liquid droplets at the
flu pandemic fears in 2006, a
needs to produce respirable
touch of a button. The technology
project plan that went from
droplets that increase – or at least
is silent, discreet and portable,
concept development to the
maintain – drug efficacy, while
making it easy to integrate
first moulded prototypes in less
eliminating the inclusion
into patients' lives.
than two months was needed.
of additional propellants.
With the development of a
two-component, single-dose,
One possible solution is
dry powder inhaler – Hovione's
TouchSpray™ technology: a
With so much information freely
TwinCaps® by TTP – for systemic
mechanism based on a nozzle
available to an unprecedented
delivery, it became possible
plate that vibrates at ultrasonic
amount of people via the
internet, patients are more aware
of the choices regarding their
medication than ever before.
While prescription treatments
and devices continue to provide
With so much information freely
invaluable benefits to users
available to an unprecedented
around the world, there is a
growing demand to provide
amount of people via the internet,
patients with the range of options
patients are more aware of the
they expect from consumer
products in their healthcare.
choices regarding their medication
In an era of choice, the trend is
likely to be a move towards the
Innovations in Pharmaceutical Technology Issue 52


development of specifi c over-the-
Understandably, some companies
2. Chow Chan C, Chow K,
counter (OTC) devices.
are looking towards the OTC market
McKay B and Fung M,
as a strategic expansion of their
Therapeutic Delivery
As patients start selecting OTC
existing medical device portfolio.
Solutions, John Wiley &
devices, an appealing design,
With inhalation-based delivery
Sons, p424, 2014. ISBN:
coupled with recognition of
devices providing so many benefi ts
their needs, is going to be
over their solid oral counterparts,
3. Chaturvedi NP and Solanki K,
exceptionally important as
it seems logical to apply these
Pulmonary drug delivery
these users become more akin
innovative technologies to OTC
system: Review, Int J App
to consumers – choosing their
medications. Such a venture could
Pharm 5(3): pp7-10, 2013
preferred device and formulation
provide patients with a valuable and
4. Olsson B et al, Pulmonary
when visiting pharmacies and
convenient alternative to traditional
drug metabolism, clearance,
dosage forms, a more effi cacious
and absorption, in Controlled
version of commonly used drugs,
Pulmonary Drug Delivery,
The OTC Market
and the opportunity for companies
Advances in Delivery Science
to capture market share along
and Technology, Smyth HDC
People frequently turn to OTCs at
and Hickey AJ (eds), Chapter 2,
the fi rst sign of disease symptoms,
rather than visiting a doctor and
Delivering an Alternative
5. TwinCaps® A simple,
requesting prescription drugs.
disposable and cost-effective
In this context, OTC medicines
Delivery of drugs via the inhalation
dry powder inhaler with
effectively provide people
route rather than the traditional
excellent performance,
with round-the-clock access
oral (swallowing) route has not
Hovione press release. Visit:
to treatment. This reliance upon
only provided effective ways of
such products is one of the
treating pulmonary diseases, but
reasons the market has become
sparked an array of innovative
so profi table: in the US alone,
technological breakthroughs.
it was valued at $17.4 billion in
By providing solutions to patient
2011 (8) – an impressive fi gure
problems, companies are ensuring
6. Blackely D et al, Drug Delivery
that could exceed $106 billion
that user needs are at the heart of
to the Lungs conference
worldwide by 2017 (9).
their concept and development
proceedings (DDL IX),
programmes, while making
It is not just patients that benefi t
signifi cant contributions to
et al, Drug Delivery
from the existence of OTCs: their
healthcare and wellbeing in
to the Lungs conference
widespread use helps to reduce
proceedings (DDL XII),
the burden on straining healthcare
systems through fewer visits to
Drug delivery technology is
8. About Money, The over-the-
local doctors, for example. One
already a sizable market, and is
counter drug industry. Visit:
estimate of the fi nancial boon
about to expand further with
that OTCs bring is a saving to
companies now looking to apply
US healthcare of $102 billion
their technologies to the OTC
annually (10).
sector, aiming to cater to patient
needs while developing robust,
9. Companies and Markets,
effective devices. With all of this
World OTC pharmaceutical
Iain McDerment, Consultant Engineer
in mind, innovative drug delivery
market 2013-2023. Visit:
at The Technology Partnership, has 19
technologies are going to be vital
years of experience in consulting and
to stand out from the competition.
drug delivery device development, and holds a BEng in Mechanical Engineering.
His specialities encompass early-stage
conceptual development through to late-stage design
1. VisionGain press release.
for manufacture. Iain has led many drug delivery device
developments from concept to high-volume production,
10. Consumer Healthcare
including the Daiichi-Sankyo Inavir inhaler. He is listed as
Products Association,
an inventor on over 25 patents, predominantly in drug
Statistics on OTC use, 2014.
delivery. Email: [email protected]
Visit: www.chpa.org/
Innovations in Pharmaceutical Technology Issue 52
Source: http://www.techpart.co.uk/files/download/2ebdb6070af7e9a
Microsoft word - 20070903 bel asx annexure to 2007 full year report - investment manager's 2007 annual portfolio report.doc
Monday, 3 September 2007 MARKET ANNOUNCEMENT Investment Manager's Report as Annexure to 2007 Full Year Report We enclose the Investment Manager's Report as an annexure to the 2007 Full Year Report dated 31 August 2007 previously lodged by the Company on ASX. For further information: Farooq Khan
Trimble dini digital level user guide
Trimble® DiNi® Digital Level Version 2.0Part Number 57345002December 2007 Corporate OfficeTrimble Navigation Limited 645 North Mary Avenue PO Box 3642 Sunnyvale, CA 94085 Geomatics and Engineering Division 5475 Kellenburger Road Dayton, Ohio 45424-1099 USA800-538-7800 (toll free in USA)